NTA JEE Main 7th January 2020 Shift 2
For the following questions answer them individually
NTA JEE Main 7th January 2020 Shift 2 - Question 31
Among statements (a) - (d), the correct ones are:
(a) Decomposition of hydrogen peroxide gives di-oxygen.
(b) Like hydrogen peroxide, compounds, such as KClO$$_3$$, Pb(NO$$_3$$) and NaNO$$_3$$ when heated liberate di-oxygen.
(c) 2-Ethylanthraquinone is useful for the industrial preparation of hydrogen peroxide.
(d) Hydrogen peroxide is used for the manufacture of sodium perborate.
NTA JEE Main 7th January 2020 Shift 2 - Question 32
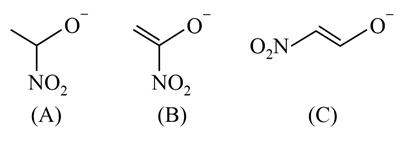
The correct order of stability for the following alkoxides is:
NTA JEE Main 7th January 2020 Shift 2 - Question 33
A chromatography column, packed with silica gel as stationary phase, was used to separate a mixture of compounds consisting of (A) benzanilide (B) aniline and (C) acetophenone. When the column is eluted with a mixture of solvents, hexane:ethyl acetate (20 : 80), the sequence of obtained compounds is:
NTA JEE Main 7th January 2020 Shift 2 - Question 34
Two open beakers one containing a solvent and the other containing a mixture of that solvent with a non volatile solute are together sealed in a container. Over time:
NTA JEE Main 7th January 2020 Shift 2 - Question 35
The equation that is incorrect is:
NTA JEE Main 7th January 2020 Shift 2 - Question 36
For the reaction $$2H_2(g) + 2NO(g) \rightarrow N_2(g) + 2H_2O(g)$$ the observed rate expression is, rate $$= k_f[NO]^2[H_2]$$. The rate expression for the reverse reaction is:
NTA JEE Main 7th January 2020 Shift 2 - Question 37
The refining method used when the metal and the impurities have low and high melting temperatures, respectively, is
NTA JEE Main 7th January 2020 Shift 2 - Question 38
In the following reactions, products (A) and (B), respectively, are:
NaOH + Cl$$_2$$ $$\rightarrow$$ (A) + side products (hot and conc.)
Ca(OH)$$_2$$ + Cl$$_2$$ $$\rightarrow$$ (B) + side products (dry)
NTA JEE Main 7th January 2020 Shift 2 - Question 39
The number of possible optical isomers for the complexes MA$$_2$$B$$_2$$ with sp$$^3$$ and dsp$$^2$$ hybridized metal atom, respectively, is:
Note: A and B are unidentate neutral and unidentate monoanionic ligands, respectively.
NTA JEE Main 7th January 2020 Shift 2 - Question 40
Among the statements (a) - (d), the incorrect ones are:
(a) Octahedral Co(III) complexes with strong field ligands have very high magnetic moments
(b) When $$\Delta_0 < P$$, the d-electron configuration of Co(III) in an octahedral complex is $$t_{eg}^4 e_g^2$$
(c) Wavelength of light absorbed by [Co(en)$$_3$$]$$^{3+}$$ is lower than that of [CoF$$_6$$]$$^{3-}$$
(d) If the $$\Delta_0$$ for an octahedral complex of Co(III) is 18,000 cm$$^{-1}$$, the $$\Delta_t$$ for its tetrahedral complex with the same ligand will be 16,000 cm$$^{-1}$$.


