NTA JEE Main 7th April 2013 Online
For the following questions answer them individually
NTA JEE Main 7th April 2013 Online - Question 41
For gaseous state, if most probable speed is denoted by $$C^*$$, average speed by $$\bar{C}$$ and root mean square speed by C, then for many molecules, what is the ratios of these speeds?
NTA JEE Main 7th April 2013 Online - Question 42
A piston filled with 0.04 mol of an ideal gas expands reversibly from 50.0 mL to 375 mL at a constant temperature of 37.0°C. As it does so, it absorbs 208 J of heat. The values of q and w for the process will be
(R = 8.314 J/mol K) (ln7.5 = 2.01)
NTA JEE Main 7th April 2013 Online - Question 43
Consider the following reaction:
$$x \ MnO_4^- + y \ C_2O_4^{2-} + zH^+ \rightarrow x \ Mn^{2+} + 2y \ CO_2 + \frac{z}{2} H_2O$$
The values of x, y and z in the reaction are, respectively:
NTA JEE Main 7th April 2013 Online - Question 44
A solution of (-)1-chloro-1-phenylethane in toluene racemises slowly in the presence of a small amount of $$SbCl_5$$, due to the formation of :
NTA JEE Main 7th April 2013 Online - Question 45
Arrange the following compounds in order of decreasing acidity :
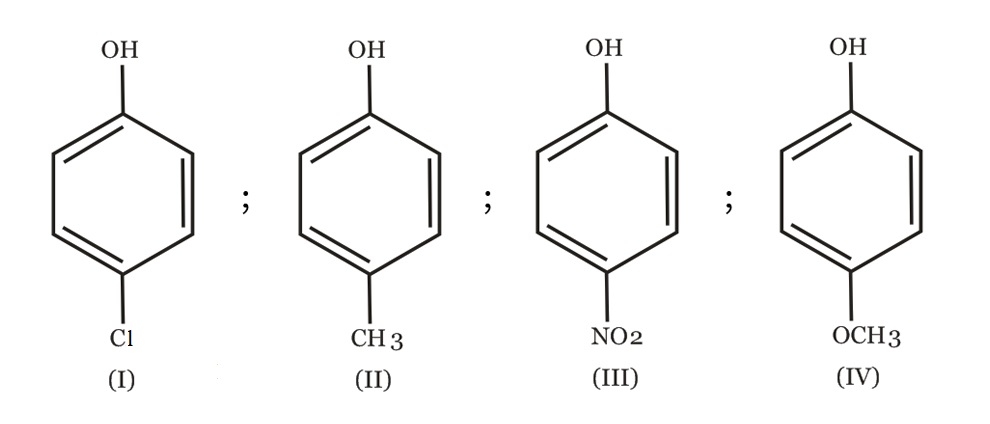
NTA JEE Main 7th April 2013 Online - Question 46
The order of stability of the following carbocations:
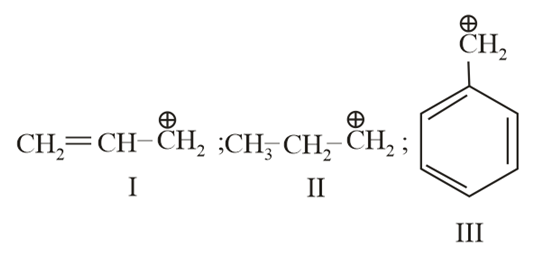
NTA JEE Main 7th April 2013 Online - Question 47
The gas leaked from a storage tank of the Union Carbide plant in Bhopal gas tragedy was :
NTA JEE Main 7th April 2013 Online - Question 48
Which of the following exists as covalent crystals in the solid state?
NTA JEE Main 7th April 2013 Online - Question 49
Four successive members of the first row of transition elements are listed below with atomic numbers. Which one of them is expected to have the highest $$E^\circ_{M^{3+}/M^{2+}}$$ value?
NTA JEE Main 7th April 2013 Online - Question 50
The rate of a reaction doubles when its temperature changes from 300K to 310K. Activation energy of such a reaction will be:
$$(R = 8.314$$ JK$$^{-1}$$ mol$$^{-1}$$ and log 2 = 0.301$$)$$


