NTA JEE Main 6th April 2023 Shift 1
For the following questions answer them individually
NTA JEE Main 6th April 2023 Shift 1 - Question 51
If 5 moles of BaCl$$_2$$ is mixed with 2 moles of Na$$_3$$PO$$_4$$, the maximum number of moles of Ba$$_3$$(PO$$_4$$)$$_2$$ formed is ______ (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 6th April 2023 Shift 1 - Question 52
The wavelength of an electron of kinetic energy $$4.50 \times 10^{-29}$$ J is ______ $$\times 10{-5}$$ m. (Nearest integer)
Given: mass of electron is $$9 \times 10{-31}$$ kg, h = $$6.6 \times 10{-34}$$ Js
789
456
123
0.-
Clear All
NTA JEE Main 6th April 2023 Shift 1 - Question 53
The number of species from the following which have square pyramidal structure is ______
PF$$_5$$, BrF$$_4^-$$, IF$$_5$$, BrF$$_5$$, XeOF$$_4$$, ICl$$_4^-$$
789
456
123
0.-
Clear All
NTA JEE Main 6th April 2023 Shift 1 - Question 54
The value of log K for the reaction A $$\rightleftharpoons$$ B at 298 K is ______. (Nearest integer)
Given: $$\Delta H° = -54.07$$ kJ mol$$^{-1}$$, $$\Delta S° = 10$$ J K$$^{-1}$$ mol$$^{-1}$$
(Taken $$2.303 \times 8.314 \times 298 = 5705$$)
789
456
123
0.-
Clear All
NTA JEE Main 6th April 2023 Shift 1 - Question 55
Consider the graph of Gibbs free energy G vs extent of reaction. The number of statement/s from the following which are true with respect to points (a), (b) and (c) is ______
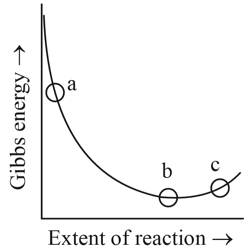
A. Reaction is spontaneous at (a) and (b)
B. Reaction is at equilibrium at point (b) and non-spontaneous at point (c)
C. Reaction is spontaneous at (a) and non-spontaneous at (c)
D. Reaction is non-spontaneous at (a) and (b)
789
456
123
0.-
Clear All
NTA JEE Main 6th April 2023 Shift 1 - Question 56
Number of bromo derivatives obtained on treating ethane with excess of Br$$_2$$ in diffused sunlight is ______
789
456
123
0.-
Clear All
NTA JEE Main 6th April 2023 Shift 1 - Question 57
Mass of Urea NH$$_2$$CONH$$_2$$ required to be dissolved in 1000 g of water in order to reduce the vapour pressure of water by 25% is ______ g. (Nearest integer)
Given: Molar mass of N, C, O and H are 14, 12, 16 and 1 g mol$$^{-1}$$ respectively.
789
456
123
0.-
Clear All
NTA JEE Main 6th April 2023 Shift 1 - Question 58
For the adsorption of hydrogen on platinum, the activation energy is 30 kJ mol$$^{-1}$$ and for the adsorption of hydrogen on nickel, the activation energy is 41.4 kJ mol$$^{-1}$$. The logarithm of the ratio of the rates of chemisorption on equal areas of the metals at 300 K is ______ (Nearest integer)
Given: ln10 = 2.3, R = 8.3 J K$$^{-1}$$ mol$$^{-1}$$
789
456
123
0.-
Clear All
NTA JEE Main 6th April 2023 Shift 1 - Question 59
In ammonium-phosphomolybdate, the oxidation state of Mo is +______
789
456
123
0.-
Clear All
NTA JEE Main 6th April 2023 Shift 1 - Question 60
Number of ambidentate ligands in a representative metal complex M(en)(SCN)$$_4$$ is ______. [en = ethylenediamine]
789
456
123
0.-
Clear All


