NTA JEE Main 4th September 2020 Shift 1
For the following questions answer them individually
NTA JEE Main 4th September 2020 Shift 1 - Question 41
An organic compound (A) (molecular formula $$C_6H_{12}O_2$$) was hydrolysed with dil. $$H_2SO_4$$ to give a carboxylic acid (B) and an alcohol (C). 'C' gives white turbidity immediately when treated with anhydrous $$ZnCl_2$$ and conc. HCl. The organic compound (A) is:
NTA JEE Main 4th September 2020 Shift 1 - Question 42
When neopentyl alcohol is heated with an acid, it slowly converted into an 85:15 mixture of alkenes A and B, respectively. What are these alkenes?
NTA JEE Main 4th September 2020 Shift 1 - Question 43
[P] on treatment with $$Br_2/FeBr_3$$ in $$CCl_4$$ produced a single isomer $$C_8H_7O_2Br$$ while heating [P] with sodalime gives toluene. The compound [P] is:
NTA JEE Main 4th September 2020 Shift 1 - Question 44
Which of the following will react with $$CHCl_3$$ + alc. KOH?
NTA JEE Main 4th September 2020 Shift 1 - Question 45
What are the functional groups present in the structure of maltose?
NTA JEE Main 4th September 2020 Shift 1 - Question 46
The mass of ammonia in grams produced when 2.8 kg of dinitrogen quantitatively reacts with 1 kg of dihydrogen is __________
789
456
123
0.-
Clear All
NTA JEE Main 4th September 2020 Shift 1 - Question 47
A 20.0 mL solution containing 0.2 g impure $$H_2O_2$$ reacts completely with 0.316 g of $$KMnO_4$$ in acid solution. The purity of $$H_2O_2$$ (in %) is __________ (mol. wt. of $$H_2O_2$$ = 34; mol. wt. of $$KMnO_4$$ = 158)
789
456
123
0.-
Clear All
NTA JEE Main 4th September 2020 Shift 1 - Question 48
At 300 K, the vapour pressure of a solution containing 1 mole of n-hexane and 3 moles of n-heptane is 550 mm of Hg. At the same temperature, if one more mole of n-heptane is added to this solution, the vapour pressure of the solution increases by 10 mm of Hg. What is the vapour pressure in mmHg of n-heptane in its pure state?
789
456
123
0.-
Clear All
NTA JEE Main 4th September 2020 Shift 1 - Question 49
If 75% of a first order reaction was completed in 90 minutes, 60% of the same reaction would be completed in approximately (in minutes) __________ (Take: log 2 = 0.30; log 2.5 = 0.40)
789
456
123
0.-
Clear All
NTA JEE Main 4th September 2020 Shift 1 - Question 50
The number of chiral centres present in [B] is __________
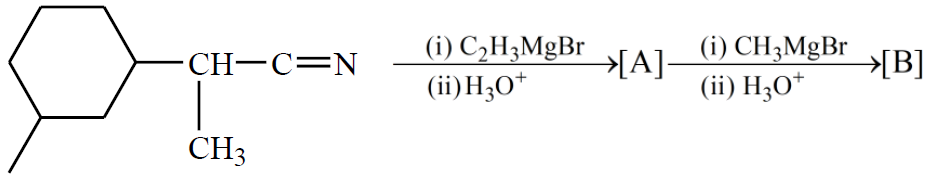
789
456
123
0.-
Clear All


