NTA JEE Main 4th September 2020 Shift 1
For the following questions answer them individually
NTA JEE Main 4th September 2020 Shift 1 - Question 31
On combustion of Li, Na and K in excess of air, the major oxides formed, respectively, are:
NTA JEE Main 4th September 2020 Shift 1 - Question 32
On heating, lead (II) nitrate gives a brown gas (A). The gas (A) on cooling changes to a colourless solid/liquid (B). (B) on heating with NO changes to a blue solid (C). The oxidation number of nitrogen in solid (C) is:
NTA JEE Main 4th September 2020 Shift 1 - Question 33
The IUPAC name of the following compound is:
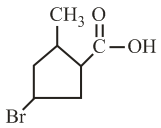
NTA JEE Main 4th September 2020 Shift 1 - Question 34
The decreasing order of reactivity of the following organic molecules towards AgNO$$_3$$ solution is:
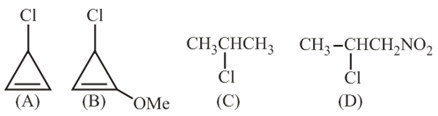
NTA JEE Main 4th September 2020 Shift 1 - Question 35
Identify the incorrect statement from the options below for the above cell:
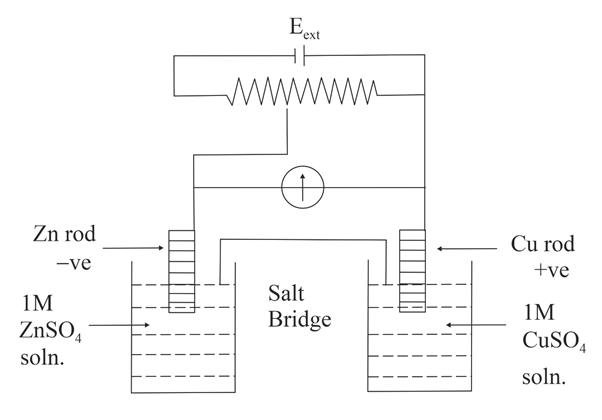
$$E^0_{Cu^{2+}|Cu} = +0.34$$ V,
$$E^0_{Zn^{2+}|Zn} = -0.76$$ V
NTA JEE Main 4th September 2020 Shift 1 - Question 36
For the equilibrium $$A \rightleftharpoons B$$, the variation of the rate of the forward (a) and reverse (b) reaction with time is given by:
NTA JEE Main 4th September 2020 Shift 1 - Question 37
Match the following:
(i) Foam (a) smoke
(ii) Gel (b) cell fluid
(iii) Aerosol (c) jellies
(iv) Emulsion (d) rubber
(e) froth
(f) milk
NTA JEE Main 4th September 2020 Shift 1 - Question 38
Among statements (a) - (d), the correct ones are:
(a) Lime stone is decomposed to CaO during the extraction of iron from its oxides.
(b) In the extraction of silver, silver is extracted as an anionic complex.
(c) Nickel is purified by Mond's process.
(d) Zr and Ti are purified by Van Arkel method.
NTA JEE Main 4th September 2020 Shift 1 - Question 39
The number of isomers possible for $$[Pt(en)(NO_2)_2]$$ is:
NTA JEE Main 4th September 2020 Shift 1 - Question 40
The pair in which both the species have the same magnetic moment (spin only) is:


