NTA JEE Main 31st January 2023 Shift 2
For the following questions answer them individually
NTA JEE Main 31st January 2023 Shift 2 - Question 51
Assume carbon burns according to following equation:
$$2C(s) + O_2(g) \rightarrow 2CO(s)$$
when 12 g carbon is burnt in 48 g of oxygen, the volume of carbon monoxide produced is ______ $$\times 10^{-1}$$ L at STP [nearest integer]
[Given: Assume CO as ideal gas, Mass of C is 12 g mol$$^{-1}$$, mass of O is 16 g mol$$^{-1}$$ and molar volume of an ideal gas at STP is 22.7 L mol$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 31st January 2023 Shift 2 - Question 52
Amongst the following, the number of species having the linear shape is
XeF$$_2$$, I$$_3^+$$, C$$_3$$O$$_2$$, I$$_3^-$$, CO$$_2$$, SO$$_2$$, BeCl$$_2$$ and BCl$$_2^\ominus$$
789
456
123
0.-
Clear All
NTA JEE Main 31st January 2023 Shift 2 - Question 53
Enthalpies of formation of CCl$$_4$$(g), H$$_2$$O(g), CO$$_2$$(g) and HCl are -105, -242, -394 and -92 kJ mol$$^{-1}$$ respectively. The magnitude of enthalpy of the reaction given below is kJmol$$^{-1}$$. (nearest integer)
$$CCl_4(g) + 2H_2O(g) \rightarrow CO_2(g) + 4HCl(g)$$
789
456
123
0.-
Clear All
NTA JEE Main 31st January 2023 Shift 2 - Question 54
At 298 K, the solubility of silver chloride in water is $$1.434 \times 10^{-3}$$ g L$$^{-1}$$. The value of $$-\log K_{sp}$$ for silver chloride is
(Given mass of Ag is 107.9 g mol$$^{-1}$$, and mass of Cl is 35.5 g mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 31st January 2023 Shift 2 - Question 55
The number of alkali metal(s), from Li, K, Cs, Rb having ionization enthalpy greater than 400 kJ mol$$^{-1}$$ and forming stable super oxide is
789
456
123
0.-
Clear All
NTA JEE Main 31st January 2023 Shift 2 - Question 56
A sample of a metal oxide has formula M$$_{0.83}$$O$$_{1.00}$$. The metal M can exist in two oxidation states +2 and +3. In the sample of M$$_{0.83}$$O$$_{1.00}$$, the percentage of metal ions existing in +2 oxidation state is %. (nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 31st January 2023 Shift 2 - Question 57
The resistivity of a 0.8M solution of an electrolyte is $$5 \times 10^{-3}$$ $$\Omega$$cm. Its molar conductivity is $$ 10^4$$ $$\Omega^{-1}$$ cm$$^2$$ mol$$^{-1}$$. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 31st January 2023 Shift 2 - Question 58
The rate constant for a first order reaction is 20 min$$^{-1}$$. The time required for the initial concentration of the reactant to reduce to its $$\dfrac{1}{32}$$ level is ______ $$\times 10^{-2}$$ min. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 31st January 2023 Shift 2 - Question 59
If the CFSE of [Ti(H$$_2$$O)$$_6$$]$$^{3+}$$ is -96.0 kJ/mol, this complex will absorb maximum at wavelength ______ nm. (nearest integer)
Assume Planck's constant (h) = $$6.4 \times 10^{-34}$$ Js, Speed of light (c) = $$3.0 \times 10^8$$ m/s and Avogadro's constant (N$$_A$$) = $$6 \times 10^{23}$$/mol.
789
456
123
0.-
Clear All
NTA JEE Main 31st January 2023 Shift 2 - Question 60
The number of molecules which gives haloform test among the following molecules is: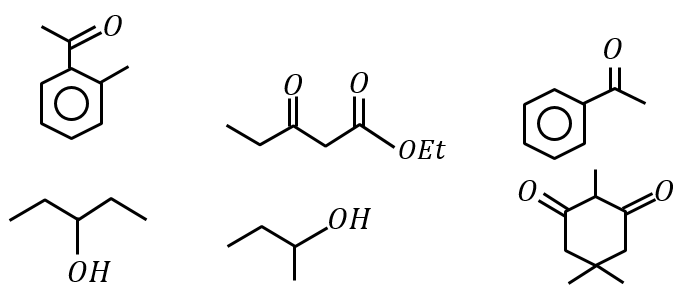
789
456
123
0.-
Clear All


