NTA JEE Main 31st January 2023 Shift 2
For the following questions answer them individually
NTA JEE Main 31st January 2023 Shift 2 - Question 41
Evaluate the following statements for their correctness.
A. The elevation in boiling point temperature of water will be same for 0.1 M NaCl and 0.1 M urea.
B. Azeotropic mixture boil without change in their composition.
C. Osmosis always takes place from hypertonic to hypotonic solution.
D. The density of 32% H$$_2$$SO$$_4$$ solution having molarity 4.09 M is approximately 1.26 g mL$$^{-1}$$.
E. A negatively charged sol is obtained when KI solution is added to silver nitrate solution.
Choose the correct answer from the options given below:
NTA JEE Main 31st January 2023 Shift 2 - Question 42
Match List I with List II
List I List II
A. Physisorption I. Single Layer Adsorption
B. Chemisorption II. 20-40 kJ mol$$^{-1}$$
C. N$$_2$$(g) + 3H$$_2$$(g) $$\xrightarrow{Fe(s)}$$ 2NH$$_3$$(g) III. Chromatography
D. Analytical Application or Adsorption IV. Heterogeneous catalysis
Choose the correct answer from the options given below:
NTA JEE Main 31st January 2023 Shift 2 - Question 43
Which one of the following statements is incorrect?
NTA JEE Main 31st January 2023 Shift 2 - Question 44
Which of the following elements have half-filled f-orbitals in their ground state?
A. Sm, B. Eu, C. Tb, D. Gd, E. Pm
(Given: atomic number Sm = 62; Eu = 63; Tb = 65; Gd = 64, Pm = 61)
Choose the correct answer from the options given below:
NTA JEE Main 31st January 2023 Shift 2 - Question 45
In the following halogenated organic compounds the one with maximum number of chlorine atoms in its structure is:
NTA JEE Main 31st January 2023 Shift 2 - Question 46
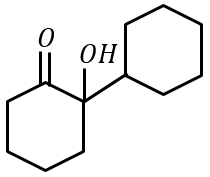
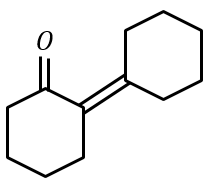
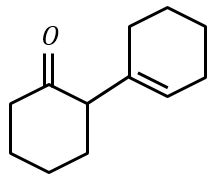
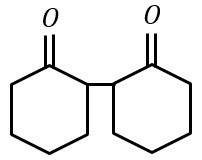
Cyclohexylamine when treated with nitrous acid yields (P). On treating (P) with PCC results in (Q). When (Q) is heated with dil. NaOH we get (R). The final product (R) is:
(1) 
(2) 
(3) 
(4) 
NTA JEE Main 31st January 2023 Shift 2 - Question 47
An organic compound [A] (C$$_4$$H$$_{11}$$N), shows optical activity and gives N$$_2$$ gas on treatment with HNO$$_2$$. The compound [A] reacts with PhSO$$_2$$Cl producing a compound which is soluble in KOH. The structure of A is:
NTA JEE Main 31st January 2023 Shift 2 - Question 48
Which of the following compounds are not used as disinfectants?
A. Chloroxylenol
B. Bithional
C. Veronal
D. Prontosil
E. Terpineol
Choose the correct answer from the options given below:
NTA JEE Main 31st January 2023 Shift 2 - Question 49
Given below are two statements:
Statement I: Upon heating a borax bead dipped in cupric sulphate in a luminous flame, the colour of the bead becomes green.
Statement II: The green colour observed is due to the formation of copper(I) metaborate.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 31st January 2023 Shift 2 - Question 50
Compound A, C$$_5$$H$$_{10}$$O$$_5$$, given a tetraacetate with AC$$_2$$O and oxidation of A with Br$$_2$$ - H$$_2$$O gives an acid, C$$_5$$H$$_{10}$$O$$_6$$. Reduction of A with HI gives isopentane. The possible structure of A is:


