NTA JEE Main 31st August 2021 Shift 1
For the following questions answer them individually
NTA JEE Main 31st August 2021 Shift 1 - Question 51
Ge (Z = 32) in its ground state electronic configuration has x completely filled orbitals with m$$_l$$ = 0. The value of x is _________.
789
456
123
0.-
Clear All
NTA JEE Main 31st August 2021 Shift 1 - Question 52
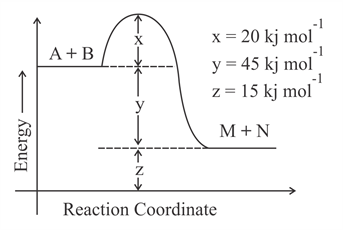
According to the following figure, the magnitude of the enthalpy change of the reaction A + B $$\rightarrow$$ M + N in kJ mol$$^{-1}$$ is equal to _________. (Integer answer)
789
456
123
0.-
Clear All
NTA JEE Main 31st August 2021 Shift 1 - Question 53
A$$_3$$B$$_2$$ is a sparingly soluble salt of molar mass M (g mol$$^{-1}$$) and solubility x g L$$^{-1}$$. The solubility product satisfies K$$_{sp}$$ = $$a\left(\frac{x}{M}\right)^5$$. The value of a is _________. (Integer answer)
789
456
123
0.-
Clear All
NTA JEE Main 31st August 2021 Shift 1 - Question 54
The number of hydrogen bonded water molecule(s) associated with stoichiometry CuSO$$_4$$.5H$$_2$$O is _________.
789
456
123
0.-
Clear All
NTA JEE Main 31st August 2021 Shift 1 - Question 55
The molarity of the solution prepared by dissolving 6.3 g of oxalic acid (H$$_2$$C$$_2$$O$$_4$$.2H$$_2$$O) in 250 mL of water in mol L$$^{-1}$$ is $$x \times 10^{-2}$$. The value of x is _________. (Nearest integer)
[Atomic mass : H : 1.0, C : 12.0, O : 16.0 J]
789
456
123
0.-
Clear All
NTA JEE Main 31st August 2021 Shift 1 - Question 56
Consider the following cell reaction Cd$$(s)$$ + Hg$$_2$$SO$$_4$$(s) + $$\frac{9}{5}$$H$$_2$$O$$(l)$$ $$\rightleftharpoons$$ CdSO$$_4$$.$$\frac{9}{5}$$H$$_2$$O$$(s)$$ + 2Hg$$(l)$$.
The value of E$$^0_{cell}$$ is 4.315 V at 25°C. If $$\Delta H°$$ = -825.2 kJ mol$$^{-1}$$, the standard entropy change $$\Delta S°$$ in J K$$^{-1}$$ is _________. (Nearest integer)
[Given : Faraday constant = 96487 C mol$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 31st August 2021 Shift 1 - Question 57
For a first order reaction, the ratio of the time for 75% completion of a reaction to the time for 50% completion is _________. (Integer answer)
789
456
123
0.-
Clear All
NTA JEE Main 31st August 2021 Shift 1 - Question 58
Consider the sulphides HgS, PbS, CuS, Sb$$_2$$S$$_3$$, As$$_2$$S$$_3$$ and CdS. Number of these sulphides soluble in 50% HNO$$_3$$ is _________.
789
456
123
0.-
Clear All
NTA JEE Main 31st August 2021 Shift 1 - Question 59
The number of halogen/(s) forming halic (V) acid is _________.
789
456
123
0.-
Clear All
NTA JEE Main 31st August 2021 Shift 1 - Question 60
The total number of reagents from those given below, that can convert nitrobenzene into aniline is _________. (Integer answer)
I. Sn - HCl
II. Sn - NH$$_4$$OH
III. Fe - HCl
IV. Zn - HCl
V. H$$_2$$ - Pd
VI. H$$_2$$ - Raney Nickel
789
456
123
0.-
Clear All


.webp)
