NTA JEE Main 3rd April 2016 Offline
For the following questions answer them individually
NTA JEE Main 3rd April 2016 Offline - Question 31
At 300 K and 1 atm, 15 mL of a gaseous hydrocarbon requires 375 mL air containing 20% $$O_2$$ by volume, for complete combustion. After combustion, the gases occupy 345 mL. Assuming that the water formed is in liquid form and the volumes were measured at the same temperature and pressure, the formula of the hydrocarbon is: (Assume complete combustion of reactant)
NTA JEE Main 3rd April 2016 Offline - Question 32
A stream of electrons from a heat filament was passed between two charge plates kept at a potential difference V esu. If e and m are charge and mass of an electron, respectively, then the value of $$\frac{h}{\lambda}$$ (where $$\lambda$$ is wavelength associated with the electron wave) is given by:
NTA JEE Main 3rd April 2016 Offline - Question 33
Which of the following atoms has the highest first ionization energy?
NTA JEE Main 3rd April 2016 Offline - Question 34
The species in which the N atom is in a state of sp hybridization is:
NTA JEE Main 3rd April 2016 Offline - Question 35
Two closed bulbs of equal volume (V) containing an ideal gas initially at pressure $$p_i$$ and temperature $$T_1$$ are connected through a narrow tube of negligible volume, as shown in the figure below. The temperature of one of the bulbs is then raised to $$T_2$$. The final pressure $$P_f$$ is:
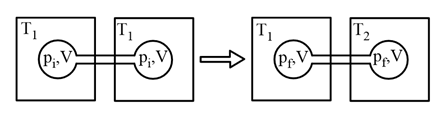
NTA JEE Main 3rd April 2016 Offline - Question 36
The heats of combustion of carbon and carbon monoxide are $$-393.5$$ and $$-283.5$$ kJ mol$$^{-1}$$, respectively. The heat of formation (in kJ) of carbon monoxide per mole is:
NTA JEE Main 3rd April 2016 Offline - Question 37
The equilibrium constant at 298 K for a reaction $$A + B \rightleftharpoons C + D$$ is 100. If the initial concentration of all the four species were 1 M each, then the equilibrium concentration of D (in mol $$L^{-1}$$) will be:
NTA JEE Main 3rd April 2016 Offline - Question 38
Which of the following statements about water is FALSE?
NTA JEE Main 3rd April 2016 Offline - Question 39
The main oxides formed on combustion of Li, Na and K in excess of air are respectively:
NTA JEE Main 3rd April 2016 Offline - Question 40
The absolute configuration of
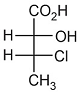
is:


