NTA JEE Main 29th June 2022 Shift 2
For the following questions answer them individually
NTA JEE Main 29th June 2022 Shift 2 - Question 51
A box contains 0.90 g of liquid water in equilibrium with water vapour at 27°C. The equilibrium vapour pressure of water at 27°C 32.0 Torr. When the volume of the box is increased, some of the liquid water evaporates to maintain the equilibrium pressure. If all the liquid water evaporates, then the volume of the box must be ______ litre. [nearest integer]
(Given: R = 0.082 L atm K$$^{-1}$$ mol$$^{-1}$$)
(Ignore the volume of the liquid water and assume water vapours behave as an ideal gas.)
789
456
123
0.-
Clear All
NTA JEE Main 29th June 2022 Shift 2 - Question 52
2.2 g of nitrous oxide (N$$_2$$O) gas is cooled at a constant pressure of 1 atm from 310 K to 270 K causing the compression of the gas from 217.1 mL to 167.75 mL. The change in internal energy of the process, $$\Delta$$U is '-x'J. The value of 'x' is ______.
[nearest integer]
(Given: atomic mass of N = 14 g mol$$^{-1}$$ and of O = 16 g mol$$^{-1}$$. Molar heat capacity of N$$_2$$O is 100 JK$$^{-1}$$ mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 29th June 2022 Shift 2 - Question 53
For the reaction given below:
CoCl$$_3$$ · xNH$$_3$$ + AgNO$$_3$$(aq) $$\rightarrow$$
If two equivalents of AgCl precipitate out, then the value of x will be ______.
789
456
123
0.-
Clear All
NTA JEE Main 29th June 2022 Shift 2 - Question 54
The number of chiral alcohol(s) with molecular formula C$$_4$$H$$_{10}$$O is ______ (Assume stereoisomers as different chiral alcohols)
789
456
123
0.-
Clear All
NTA JEE Main 29th June 2022 Shift 2 - Question 55
Elevation in boiling point for 1.5 molal solution of glucose in water is 4 K. The depression in freezing point for 4.5 molal solution of glucose in water is 4 K. The ratio of molal elevation constant to molal depression constant (K$$_b$$/K$$_f$$) is
789
456
123
0.-
Clear All
NTA JEE Main 29th June 2022 Shift 2 - Question 56
The cell potential for the given cell at 298 K Pt|H$$_2$$(g, 1 bar)|H$$^+$$(aq)||Cu$$^{2+}$$(aq)|Cu(s) is 0.31 V. The pH of the acidic solution is found to be 3, whereas the concentration of Cu$$^{2+}$$ is $$10^x$$ M. The value of x is ______.
(Given: $$E^\ominus_{Cu^{2+}/Cu} = 0.34$$ V and $$\frac{2.303RT}{F} = 0.06$$ V)
789
456
123
0.-
Clear All
NTA JEE Main 29th June 2022 Shift 2 - Question 57
The equation $$k = (6.5 \times 10^{12} s^{-1})e^{-26000 K/T}$$ is followed for the decomposition of compound A. The activation energy for the reaction is ______ KJ mol$$^{-1}$$. [nearest integer]
(Given: R = 8.314 J K$$^{-1}$$ mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 29th June 2022 Shift 2 - Question 58
Spin only magnetic moment of $$[MnBr_6]^{4-}$$ is ______ B.M. (round off to the closest integer)
789
456
123
0.-
Clear All
NTA JEE Main 29th June 2022 Shift 2 - Question 59
In the given reaction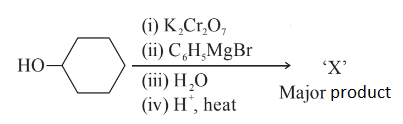
the number of sp$$^2$$ hybridised carbon (s) in compound 'X' is ______.
789
456
123
0.-
Clear All
NTA JEE Main 29th June 2022 Shift 2 - Question 60
In the given reaction,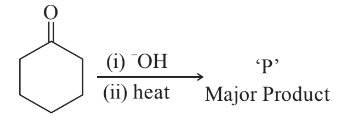
The number of $$\pi$$ electrons present in the product 'P' is ______.
789
456
123
0.-
Clear All


