NTA JEE Main 29th June 2022 Shift 2
For the following questions answer them individually
NTA JEE Main 29th June 2022 Shift 2 - Question 41
42.12% (w/v) solution of NaCl causes precipitation of a certain sol in 10 hours. The coagulating value of NaCl for the sol is
[Given: Molar mass: Na = 23.0 g mol$$^{-1}$$; Cl = 35.5 g mol$$^{-1}$$]
NTA JEE Main 29th June 2022 Shift 2 - Question 42
Match List I with List II.
List I With Ore List II Composition
(A) Siderite (I) FeCO$$_3$$
(B) Malachite (II) CuCO$$_3$$ · Cu(OH)$$_2$$
(C) Sphalerite (III) ZnS
(D) Calamine (IV) ZnCO$$_3$$
Choose the correct answer from the options given below:
NTA JEE Main 29th June 2022 Shift 2 - Question 43
Given below are two statements.
Statement I: In CuSO$$_4$$ · 5H$$_2$$O, Cu - O bonds are present.
Statement II: In CuSO$$_4$$ · 5H$$_2$$O, ligands coordinating with Cu (II) ion are O-and S-based ligands.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Main 29th June 2022 Shift 2 - Question 44
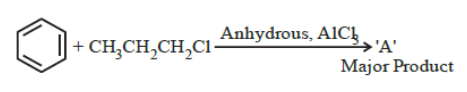
The stable carbocation formed in the above reaction is:
NTA JEE Main 29th June 2022 Shift 2 - Question 45
Two isomers (A) and (B) with Molar mass 184 g/mol and elemental composition C, 52.2%; H, 49% and Br 42.9% gave benzoic acid and p-bromobenzoic acid, respectively on oxidation with KMnO$$_4$$. Isomer 'A' is optically active and gives a pale yellow precipitate when warmed with alcoholic AgNO$$_3$$. Isomer 'A' and 'B' are, respectively:
NTA JEE Main 29th June 2022 Shift 2 - Question 46
In Friedel-Crafts alkylation of aniline, one gets:
NTA JEE Main 29th June 2022 Shift 2 - Question 47
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: Dacron is an example of polyester polymer.
Reason R: Dacron is made up of ethylene glycol and terephthalic acid monomers.
In the light of the above statements, choose the most appropriate answer from the options given below.
NTA JEE Main 29th June 2022 Shift 2 - Question 48
The mixture of chloroxylenol and terpineol is an example of:
NTA JEE Main 29th June 2022 Shift 2 - Question 49
A white precipitate was formed when BaCl$$_2$$ was added to water extract of an inorganic salt. Further, a gas 'X' with characteristic odour was released when the formed white precipitate was dissolved in dilute HCI. The anion present in the inorganic salt is:
NTA JEE Main 29th June 2022 Shift 2 - Question 50
The structure of protein that is unaffected by heating is:


