NTA JEE Main 29th June 2022 Shift 1
For the following questions answer them individually
NTA JEE Main 29th June 2022 Shift 1 - Question 51
Geraniol, a volatile organic compound, is a component of rose oil. The density of the vapour is 0.46 gL$$^{-1}$$ at 257°C and 100 mmHg. The molar mass of geraniol is ______ (Nearest Integer)
[Given R = 0.082 L atm K$$^{-1}$$ mol$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 29th June 2022 Shift 1 - Question 52
17.0 g of NH$$_3$$ completely vapourises at $$-33.42$$°C and 1 bar pressure and the enthalpy change in the process is 23.4 kJ mol$$^{-1}$$. The enthalpy change for the vapourisation of 85 g of NH$$_3$$ under the same conditions is ______ kJ.
789
456
123
0.-
Clear All
NTA JEE Main 29th June 2022 Shift 1 - Question 53
Kjeldahl's method was used for the estimation of nitrogen in an organic compound. The ammonia evolved from 0.55 g of the compound neutralised 12.5 mL of 1M H$$_2$$SO$$_4$$ solution. The percentage of nitrogen in the compound is ______ (Nearest Integer)
789
456
123
0.-
Clear All
NTA JEE Main 29th June 2022 Shift 1 - Question 54
Observe structures of the following compounds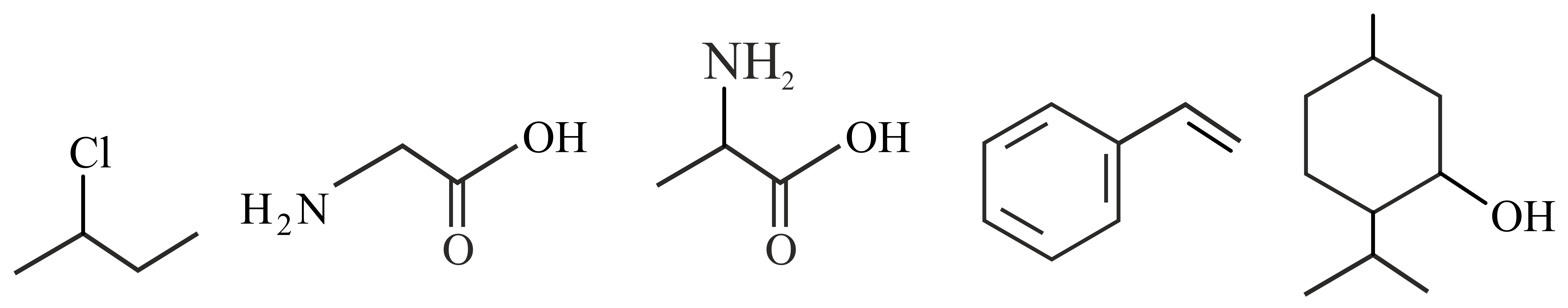
The total number of structures/compounds which possess asymmetric carbon atoms is
789
456
123
0.-
Clear All
NTA JEE Main 29th June 2022 Shift 1 - Question 55
1.2 mL of acetic acid is dissolved in water to make 2.0 L of solution. The depression in freezing point observed for this strength of acid is 0.0198°C. The percentage of dissociation of the acid is ______ (Nearest integer)
[Given: Density of acetic acid is 1.02 g mL$$^{-1}$$ Molar mass of acetic acid is 60 g mol$$^{-1}$$, K$$_f$$(H$$_2$$O) = 1.85 K kg mol$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 29th June 2022 Shift 1 - Question 56
A dilute solution of sulphuric acid is electrolysed using a current of 0.10 A for 2 hours to produce hydrogen and oxygen gas. The total volume of gases produced at STP is ______ cm$$^3$$. (Nearest integer) [Given: Faraday constant F = 96500 C mol$$^{-1}$$ at STP, molar volume of an ideal gas is 22.7 L mol$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 29th June 2022 Shift 1 - Question 57
The activation energy of one of the reactions in a biochemical process is 532611 J mol$$^{-1}$$. When the temperature falls from 310 K to 300 K, the change in rate constant observed is k$$_{300}$$ = x $$\times$$ 10$$^{-3}$$k$$_{310}$$. The value of x is ______.
[Given: ln 10 = 2.3 R = 8.3 J K$$^{-1}$$ mol$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 29th June 2022 Shift 1 - Question 58
The number of terminal oxygen atoms present in the product B obtained from the following reaction is ______.
FeCr$$_2$$O$$_4$$ + Na$$_2$$CO$$_3$$ + O$$_2$$ $$\rightarrow$$ A + Fe$$_2$$O$$_3$$ + CO$$_2$$
A + H$$^+$$ $$\rightarrow$$ B + H$$_2$$O + Na$$^+$$
789
456
123
0.-
Clear All
NTA JEE Main 29th June 2022 Shift 1 - Question 59
An acidified manganate solution undergoes disproportionation reaction. The spin-only magnetic moment value of the product having manganese in higher oxidation state is ______ B.M. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 29th June 2022 Shift 1 - Question 60
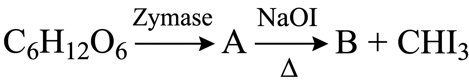
The number of carbon atoms present in the product B is
789
456
123
0.-
Clear All


