NTA JEE Main 28th June 2022 Shift 2
For the following questions answer them individually
NTA JEE Main 28th June 2022 Shift 2 - Question 51
The complete combustion of 0.492 g of an organic compound containing 'C', 'H' and 'O' gives 0.793 g of CO$$_2$$ and 0.442 g of H$$_2$$O. The percentage of oxygen composition in the organic compound is ______ (nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 28th June 2022 Shift 2 - Question 52
100 g of an ideal gas is kept in a cylinder of 416 L volume at 27°C under 1.5 bar pressure. The molar mass of the gas is ______ g mol$$^{-1}$$. (Nearest integer)
(Given: R = 0.083 L bar K$$^{-1}$$ mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 28th June 2022 Shift 2 - Question 53
For combustion of one mole of magnesium in an open container at 300 K and 1 bar pressure, $$\Delta_C H^\ominus = -601.70$$ kJ mol$$^{-1}$$, the magnitude of change in internal energy for the reaction is ______ kJ. (Nearest integer)
(Given: R = 8.3 J K$$^{-1}$$ mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 28th June 2022 Shift 2 - Question 54
0.01 M KMnO$$_4$$ solution was added to 20.0 mL of 0.05 M Mohr's salt solution through a burette. The initial reading of 50 mL burette is zero. The volume of KMnO$$_4$$ solution left in the burette after the end point is ______ mL. (nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 28th June 2022 Shift 2 - Question 55
For the given reactions
$$Sn^{2+} + 2e^- \rightarrow Sn$$
$$Sn^{4+} + 4e^- \rightarrow Sn$$
the electrode potentials are; $$E^\circ_{Sn^{2+}/Sn} = -0.140$$ V and $$E^\circ_{Sn^{4+}/Sn} = 0.010$$ V. The magnitude of standard electrode potential for $$Sn^{4+}/Sn^{2+}$$ i.e. $$E^\circ_{Sn^{4+}/Sn^{2+}}$$ is ______ $$\times 10^{-2}$$ V (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 28th June 2022 Shift 2 - Question 56
A radioactive element has a half life of 200 days. The percentage of original activity remaining after 83 days is ______ (Nearest integer)
(Given: antilog 0.125 = 1.333, antilog 0.693 = 4.93)
789
456
123
0.-
Clear All
NTA JEE Main 28th June 2022 Shift 2 - Question 57
$$[Fe(CN)_6]^{4-}$$
$$[Fe(CN)_6]^{3-}$$
$$[Ti(CN)_6]^{3-}$$
$$[Ni(CN)_4]^{2-}$$
$$[Co(CN)_6]^{3-}$$
Among the given complexes, number of paramagnetic complexes is
789
456
123
0.-
Clear All
NTA JEE Main 28th June 2022 Shift 2 - Question 58
(a) CoCl$$_3$$ · 4NH$$_3$$,
(b) CoCl$$_3$$ · 5NH$$_3$$,
(c) CoCl$$_3$$ · 6NH$$_3$$ and
(d) CoCl(NO$$_3$$)$$_2$$ · 5NH$$_3$$
Number of complex(es) which will exist in cis-trans form is/are
789
456
123
0.-
Clear All
NTA JEE Main 28th June 2022 Shift 2 - Question 59
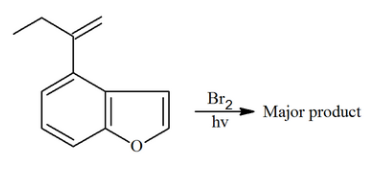
The major product of the following reaction contains ______ bromine atom(s).
789
456
123
0.-
Clear All
NTA JEE Main 28th June 2022 Shift 2 - Question 60
2.5 g of protein containing only glycine (C$$_2$$H$$_5$$NO$$_2$$) is dissolved in water to make 500 mL of solution. The osmotic pressure of this solution at 300 K is found to be $$5.03 \times 10^{-3}$$ bar. The total number of glycine units present in the protein is
(Given: R = 0.083 L bar K$$^{-1}$$ mol$$^{-1}$$)
789
456
123
0.-
Clear All


