NTA JEE Main 28th June 2022 Shift 2
For the following questions answer them individually
NTA JEE Main 28th June 2022 Shift 2 - Question 31
Compound A contains 8.7% Hydrogen, 74% Carbon and 17.3% Nitrogen. The molecular formula of the compound is, Given: Atomic masses of C, H and N are 12, 1 and 14 amu respectively. The molar mass of the compound A is 162 g mol$$^{-1}$$.
NTA JEE Main 28th June 2022 Shift 2 - Question 32
Consider the following statements:
(A) The principal quantum number 'n' is a positive integer with values of 'n' = 1, 2, 3, ...
(B) The azimuthal quantum number 'l' for a given 'n' (principal quantum number) can have values as 'l' = 0, 1, 2, ... n has $$(2n + 1)$$ values.
(C) Magnetic orbital quantum number 'm' for a particular 'l' (azimuthal quantum number) has $$(2l + 1)$$ values.
(D) $$\pm \frac{1}{2}$$ are the two possible orientations of electron spin.
(E) For $$l = 5$$, there will be a total of 9 orbital.
Which of the above statements are correct?
NTA JEE Main 28th June 2022 Shift 2 - Question 33
Match List-I with List-II
List-I List-II
(A) Cl$$_2$$O$$_7$$ (I) Amphoteric
(B) Na$$_2$$O (II) Basic
(C) Al$$_2$$O$$_3$$ (III) Neutral
(D) N$$_2$$O (IV) Acidic
Choose the correct answer from the options given below
NTA JEE Main 28th June 2022 Shift 2 - Question 34
In the structure of SF$$_4$$, the lone pair of electrons on S is in.
NTA JEE Main 28th June 2022 Shift 2 - Question 35
A student needs to prepare a buffer solution of propanoic acid and its sodium salt with pH 4. The ratio of $$\frac{[CH_3CH_2COO^-]}{[CH_3CH_2COOH]}$$ required to make buffer is Given: $$K_a(CH_3CH_2COOH) = 1.3 \times 10^{-5}$$
NTA JEE Main 28th June 2022 Shift 2 - Question 36
Hydrogen has three isotopes: protium ($$^1$$H), deuterium ($$^2$$H or D) and tritium ($$^3$$H or T). They have nearly same chemical properties but different physical properties. They differ in
NTA JEE Main 28th June 2022 Shift 2 - Question 37
Among the following, basic oxide is
NTA JEE Main 28th June 2022 Shift 2 - Question 38
The correct IUPAC name of the following compound is
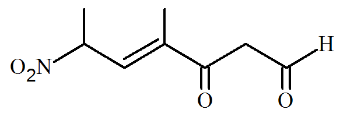
NTA JEE Main 28th June 2022 Shift 2 - Question 39
Correct statement about photo-chemical smog is
NTA JEE Main 28th June 2022 Shift 2 - Question 40
Match List-I with List-II
List-I List-II
(A) negatively charged sol (I) Fe$$_2$$O$$_3$$ · xH$$_2$$O
(B) macromolecular colloid (II) CdS sol
(C) positively charged sol (III) Starch
(D) Cheese (IV) a gel
Choose the correct answer from the options given below


