NTA JEE Main 27th June 2022 Shift 2
For the following questions answer them individually
NTA JEE Main 27th June 2022 Shift 2 - Question 51
$$116$$ g of a substance upon dissociation reaction, yields $$7.5$$ g of hydrogen, $$60$$ g of oxygen and $$48.5$$ g of carbon. Given that the atomic masses of H, O and C are 1, 16 and 12, respectively. The data agrees with how many formulae of the following?
A. $$CH_3COOH$$
B. HCHO
C. $$CH_3OOCH_3$$
D. $$CH_3CHO$$
789
456
123
0.-
Clear All
NTA JEE Main 27th June 2022 Shift 2 - Question 52
Consider the following set of quantum numbers.
A: n=3, l=3, m$$_{1}$$=-3
B: n=3, l=2, m$$_{1}$$=-2
C: n=2, l=1, m$$_{1}$$=+1
D: n=2, l=2, m$$_{1}$$=+2
The number of correct sets of quantum numbers is ______
789
456
123
0.-
Clear All
NTA JEE Main 27th June 2022 Shift 2 - Question 53
When 5 moles of He gas expand isothermally and reversibly at $$300$$ K from $$10$$ litre to $$20$$ litre, the magnitude of the maximum work obtained is ______ J. [nearest integer] (Given : $$R = 8.3$$ J K$$^{-1}$$ mol$$^{-1}$$ and $$\log 2 = 0.3010$$)
789
456
123
0.-
Clear All
NTA JEE Main 27th June 2022 Shift 2 - Question 54
pH value of $$0.001$$ M NaOH solution is ______
789
456
123
0.-
Clear All
NTA JEE Main 27th June 2022 Shift 2 - Question 55
BeO reacts with HF in presence of ammonia to give [A] which on thermal decomposition produces [B] and ammonium fluoride. Oxidation state of Be in [A] is ______
789
456
123
0.-
Clear All
NTA JEE Main 27th June 2022 Shift 2 - Question 56
$$0.25$$ g of an organic compound containing chlorine gave $$0.40$$ g of silver chloride in Carius estimation. The percentage of chlorine present in the compound is ______ [in nearest integer]
(Given: Molar mass of Ag is $$108$$ g mol$$^{-1}$$ and that of Cl is $$35.5$$ g mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 27th June 2022 Shift 2 - Question 57
A solution containing $$2.5 \times 10^{-3}$$ kg of a solute dissolved in $$75 \times 10^{-3}$$ kg of water boils at $$373.535$$ K. The molar mass of the solute is ______ mol$$^{-1}$$. [nearest integer] (Given : $$K_b(H_2O) = 0.52$$ K kg mol$$^{-1}$$ and boiling point of water $$= 373.15$$ K)
789
456
123
0.-
Clear All
NTA JEE Main 27th June 2022 Shift 2 - Question 58
For the reaction taking place in the cell:
$$Pt(s)|H_2(g)|H^+(aq) || Ag^+(aq)|Ag(s)$$
$$E_{cell} = +0.5332$$ V.
The value of $$\Delta_r G^\circ$$ is ______ kJ mol$$^{-1}$$. (in nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 27th June 2022 Shift 2 - Question 59
It has been found that for a chemical reaction with rise in temperature by $$9$$ K the rate constant gets doubled. Assuming a reaction to be occurring at $$300$$ K, the value of activation energy is found to be ______ kJ mol$$^{-1}$$. [nearest integer] (Given $$\ln 10 = 2.3, R = 8.3$$ J K$$^{-1}$$ mol$$^{-1}$$, $$\log 2 = 0.30$$)
789
456
123
0.-
Clear All
NTA JEE Main 27th June 2022 Shift 2 - Question 60
A Freundlich adsorption isotherm graph is shown.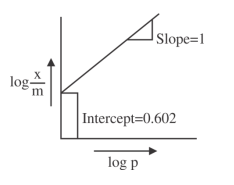
If the initial pressure of a gas is $$0.03$$ atm, the mass of the gas adsorbed per gram of the adsorbent is ______ $$\times 10^{-2}$$ g
789
456
123
0.-
Clear All


