NTA JEE Main 27th July 2021 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 27th July 2021 Shift 2 - Chemistry - Question 31
If the Thomson model of the atom was correct, then the result of Rutherford's gold foil experiment would have been:
NTA JEE Main 27th July 2021 Shift 2 - Chemistry - Question 32
The CORRECT order of first ionisation enthalpy is:
NTA JEE Main 27th July 2021 Shift 2 - Chemistry - Question 33
The number of neutrons and electrons, respectively, present in the radioactive isotope of hydrogen is:
NTA JEE Main 27th July 2021 Shift 2 - Chemistry - Question 34
Match List - I with List II:
List-I List-II
a. Li i. photoelectric cell
b. Na ii. absorbent of CO$$_2$$
c. K iii. coolant in fast breeder nuclear reactor
d. Cs iv. treatment of cancer
v. bearings for motor engines
Choose the correct answer from the options given below:
NTA JEE Main 27th July 2021 Shift 2 - Chemistry - Question 35
Which one of the following set of elements can be detected using sodium fusion extract?
NTA JEE Main 27th July 2021 Shift 2 - Chemistry - Question 36
Given below are two statements:
Statement I : Hyperconjugation is a permanent effect.
Statement II : Hyperconjugation in ethyl cation $$\left(CH_3 - \overset{+}{CH_2}\right)$$ involves the overlapping of $$C_{sp^2} - H_{1s}$$ bond with empty 2p orbital of other carbon.
Choose the correct option:
NTA JEE Main 27th July 2021 Shift 2 - Chemistry - Question 37
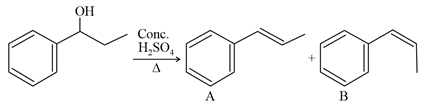
Consider the above reaction, and choose the correct statement:
NTA JEE Main 27th July 2021 Shift 2 - Chemistry - Question 38
The correct sequence of correct reagents for the following transformation is:-
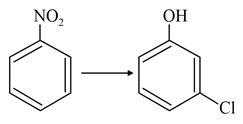
NTA JEE Main 27th July 2021 Shift 2 - Chemistry - Question 39
Match List - I with List - II:
List - I (compound) List - II (effect/affected species)
a. Carbon monoxide i. Carcinogenic
b. Sulphur dioxide ii. Metabolized by pyrus plants
c. Polychlorinated biphenyls iii. Haemoglobin
d. Oxides of Nitrogen iv. Stiffness of flower buds
Choose the correct answer from the options given below:
NTA JEE Main 27th July 2021 Shift 2 - Chemistry - Question 40
Select the correct statements.
(A) Crystalline solids have long range order.
(B) Crystalline solids are isotropic.
(C) Amorphous solid are sometimes called pseudo solids.
(D) Amorphous solids soften over a range of temperatures.
(E) Amorphous solids have a definite heat of fusion.
Choose the most appropriate answer from the options given below.
NTA JEE Main 27th July 2021 Shift 2 - Chemistry - Question 41
Given below are two statement: one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A : SO$$_2$$(g) is adsorbed to a large extent than H$$_2$$(g) on activated charcoal.
Reason R : SO$$_2$$(g) has a higher critical temperature than H$$_2$$(g)
In the light of the above statements, choose the most appropriate answer from the options given below.
NTA JEE Main 27th July 2021 Shift 2 - Chemistry - Question 42
The addition of silica during the extraction of copper from its sulphide ore:-
NTA JEE Main 27th July 2021 Shift 2 - Chemistry - Question 43
Number of Cl = O bonds in chlorous acid, chloric acid and perchloric acid respectively are:
NTA JEE Main 27th July 2021 Shift 2 - Chemistry - Question 44
Given below are two statements:
Statement I: $$[Mn(CN)_6]^{3-}$$, $$[Fe(CN)_6]^{3-}$$ and $$[Co(C_2O_4)_3]^{3-}$$ are d$$^2$$sp$$^3$$ hybridised.
Statement II: $$[MnCl_6]^{3-}$$ and $$[FeF_6]^{3-}$$ are paramagnetic and have 4 and 5 unpaired electrons, respectively.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Main 27th July 2021 Shift 2 - Chemistry - Question 45
What is A in the following reaction?
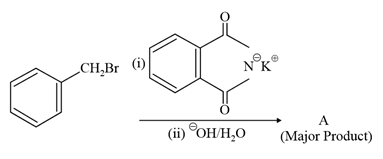
NTA JEE Main 27th July 2021 Shift 2 - Chemistry - Question 46
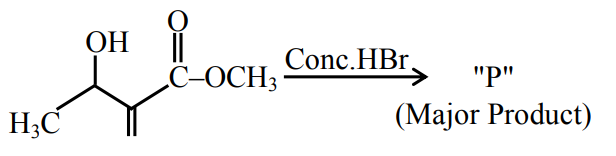
Consider the above reaction, the major product P formed is:
NTA JEE Main 27th July 2021 Shift 2 - Chemistry - Question 47
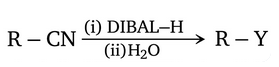
Consider the above reaction and identify "Y"
NTA JEE Main 27th July 2021 Shift 2 - Chemistry - Question 48
Given below are two statements:
Statement I : Penicillin is a bacteriostatic type antibiotic.
Statement II : The general structure of Penicillin is:
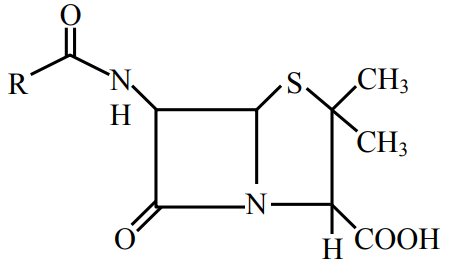
Choose the correct option:
NTA JEE Main 27th July 2021 Shift 2 - Chemistry - Question 49
To an aqueous solution containing ions such as Al$$^{3+}$$, Zn$$^{2+}$$, Ca$$^{2+}$$, Fe$$^{3+}$$, Ni$$^{2+}$$, Ba$$^{2+}$$ and Cu$$^{2+}$$ was added conc. HCl, followed by H$$_2$$S. The total number of cations precipitated during this reaction is/are:
NTA JEE Main 27th July 2021 Shift 2 - Chemistry - Question 50
Compound A gives D-Galactose and D-Glucose on hydrolysis. The compound A is:
NTA JEE Main 27th July 2021 Shift 2 - Chemistry - Question 51
$$2SO_2(g) + O_2(g) \rightarrow 2SO_3(g)$$
The above reaction is carried out in a vessel starting with partial pressure $$P_{SO_2} = 250$$ m bar, $$P_{O_2} = 750$$ m bar and $$P_{SO_3} = 0$$ bar. When the reaction is complete, the total pressure in the reaction vessel is _________ m bar. (Round off to the nearest integer).
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2021 Shift 2 - Chemistry - Question 52
The total number of electrons in all bonding molecular orbitals of $$O_2^{2-}$$ is _________ (Round off to the nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2021 Shift 2 - Chemistry - Question 53
When 400 mL of 0.2 M H$$_2$$SO$$_4$$ solution is mixed with 600 mL of 0.1 M NaOH solution, the increase in temperature of the final solution is _________ $$\times 10^{-2}$$ K. (Round off to the nearest integer).
[Use: H$$^+$$(aq) + OH$$^-$$(aq) $$\rightarrow$$ H$$_2$$O: $$\Delta_r H = -57.1$$ kJ mol$$^{-1}$$
Specific heat of H$$_2$$O = 4.18 J K$$^{-1}$$ g$$^{-1}$$, density of H$$_2$$O = 1.0 g cm$$^{-3}$$
Assume no change in volume of solution on mixing.]
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2021 Shift 2 - Chemistry - Question 54
The equilibrium constant for the reaction $$A(s) \rightleftharpoons M(s) + \frac{1}{2}O_2(g)$$ is $$K_p = 4$$. At equilibrium, the partial pressure of O$$_2$$ is _________ atm. (Round off to the nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2021 Shift 2 - Chemistry - Question 55
10.0 mL of 0.05 M KMnO$$_4$$ solution was consumed in a titration with 10.0 mL of given oxalic acid dihydrate solution. The strength of given oxalic acid solution is _________ $$\times 10^{-2}$$ g/L. (Round off to the nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2021 Shift 2 - Chemistry - Question 56
The dihedral angle in staggered form of Newmann's projection of 1, 1, 1-Trichloro ethane is _________ degree. (Round off to the nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2021 Shift 2 - Chemistry - Question 57
In a solvent 50% of an acid HA dimerizes and the rest dissociates. The van't Hoff factor of the acid is _________ $$\times 10^{-2}$$ (Round off to the nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2021 Shift 2 - Chemistry - Question 58
For the cell Cu(s)|Cu$$^{2+}$$(aq)(0.1M)||Ag$$^+$$(aq)(0.01M)|Ag(s) the cell potential E$$_1$$ = 0.3095 V. For the cell Cu(s)|Cu$$^{2+}$$(aq)(0.01M)||Ag$$^+$$(aq)(0.001M)|Ag(s) the cell potential = $$x \times 10^{-2}$$ V. Find value of x.
(Round off to the Nearest Integer).
[Use: $$\frac{2.303 RT}{F} = 0.059$$ J]
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2021 Shift 2 - Chemistry - Question 59
For the first order reaction A $$\rightarrow$$ 2B, 1 mole of reactant A gives 0.2 moles of $$B$$ after 100 minutes. The half life of the reaction is _________ min. (Round off to nearest integer).
[$$Use: ln 2 = 0.69, ln 10 = 2.3$$
Properties of logarithms: $$\ln x^y = y \ln x$$
$$\ln\left(\frac{x}{y}\right)=\ln x-\ln y$$]
(Round off to the nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2021 Shift 2 - Chemistry - Question 60
3 moles of metal complex with formula Co(en)$$_2$$Cl$$_3$$ gives 3 moles of silver chloride on treatment with excess of silver nitrate. The secondary valency of Co in the complex is _________ (Round off to the nearest integer)
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




