NTA JEE Main 27th July 2021 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 27th July 2021 Shift 1 - Chemistry - Question 31
Given below are two statements:
Statement I : Rutherford's gold foil experiment cannot explain the line spectrum of hydrogen atom.
Statement II : Bohr's model of hydrogen atom contradicts Heisenberg's uncertainty principle.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 27th July 2021 Shift 1 - Chemistry - Question 32
Match List - I with List - II:
List-I List-II
(a) NaOH (i) Acidic
(b) Be(OH)$$_2$$ (ii) Basic
(c) Ca(OH)$$_2$$ (iii) Amphoteric
(d) B(OH)$$_3$$
(e) Al(OH)$$_3$$
Choose the most appropriate answer from the options given below:
NTA JEE Main 27th July 2021 Shift 1 - Chemistry - Question 33
Given below are two statements: One is labelled as Assertion A and the other labelled as Reason R.
Assertion A : Lithium halides are somewhat covalent in nature.
Reason R : Lithium possess high polarisation capability.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 27th July 2021 Shift 1 - Chemistry - Question 34
The oxidation states of $$P$$ in H$$_4$$P$$_2$$O$$_7$$, H$$_4$$P$$_2$$O$$_5$$ and H$$_4$$P$$_2$$O$$_6$$, respectively, are:
NTA JEE Main 27th July 2021 Shift 1 - Chemistry - Question 35
Given below are two statements:
Statement I : Aniline is less basic than acetamide.
Statement II : In aniline, the lone pair of electrons on nitrogen atom is delocalised over benzene ring due to resonance and hence less available to a proton.
Choose the most appropriate option:
NTA JEE Main 27th July 2021 Shift 1 - Chemistry - Question 36
Staggered and eclipsed conformers of ethane are:
NTA JEE Main 27th July 2021 Shift 1 - Chemistry - Question 37
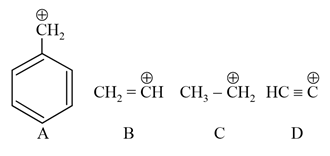
The correct order of stability of given carbocation is:
NTA JEE Main 27th July 2021 Shift 1 - Chemistry - Question 38
Presence of which reagent will affect the reversibility of the following reaction, and change it to a irreversible reaction:
$$CH_4 + I_2 \underset{\text{Reversible}}{\overset{h\nu}{\rightleftharpoons}} CH_3 - I + HI$$
NTA JEE Main 27th July 2021 Shift 1 - Chemistry - Question 39
Which one of the following statements is NOT correct?
NTA JEE Main 27th July 2021 Shift 1 - Chemistry - Question 40
The parameters of the unit cell of a substance are a = 2.5, b = 3.0, c = 4.0, $$\alpha$$ = 90°, $$\beta$$ = 120°, $$\gamma$$ = 90°. The crystal system of the substance is:
NTA JEE Main 27th July 2021 Shift 1 - Chemistry - Question 41
For a reaction of order n, the unit of the rate constant is:
NTA JEE Main 27th July 2021 Shift 1 - Chemistry - Question 42
The statement that is INCORRECT about Ellingham diagram is
NTA JEE Main 27th July 2021 Shift 1 - Chemistry - Question 43
The product obtained from the electrolytic oxidation of acidified sulphate solutions, is:
NTA JEE Main 27th July 2021 Shift 1 - Chemistry - Question 44
The type of hybridisation and magnetic property of the complex $$[MnCl_6]^{3-}$$, respectively, are:
NTA JEE Main 27th July 2021 Shift 1 - Chemistry - Question 45
The number of geometrical isomers found in the metal complexes $$[PtCl_2(NH_3)_2]$$, $$[Ni(CO)_4]$$, $$[Ru(H_2O)_3Cl_3]$$ and $$[CoCl_2(NH_3)_4]^+$$ respectively, are:
NTA JEE Main 27th July 2021 Shift 1 - Chemistry - Question 46
Which one of the following compounds will give orange precipitate when treated with 2,4-dinitrophenyl hydrazine?
NTA JEE Main 27th July 2021 Shift 1 - Chemistry - Question 47
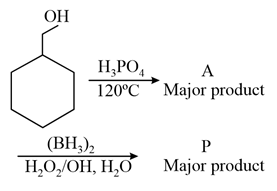
Consider the above reaction and identify the Product P:
NTA JEE Main 27th July 2021 Shift 1 - Chemistry - Question 48
Match List-I (Drug) with List-II (Class of Drug):
(a) Furacin (i) Antibiotic
(b) Arsphenamine (ii) Tranquilizers
(c) Dimetone (iii) Antiseptic
(d) Valium (iv) Synthetic antihistamines
Choose the most appropriate match:
NTA JEE Main 27th July 2021 Shift 1 - Chemistry - Question 49
Which one among the following chemical tests is used to distinguish monosaccharide from disaccharide?
NTA JEE Main 27th July 2021 Shift 1 - Chemistry - Question 50
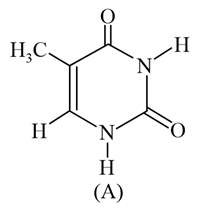
The compound 'A' is a complementary base of _________ in DNA strands.
NTA JEE Main 27th July 2021 Shift 1 - Chemistry - Question 51
The density of NaOH solution is 1.2 g cm$$^{-3}$$. The molality of this solution is _________ m (Round off to the Nearest Integer):
[Use: Atomic masses: Na : 23.0u, O : 16.0u, H : 1.0u
Density of H$$_2$$O : 1.0 g cm$$^{-3}$$]
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2021 Shift 1 - Chemistry - Question 52
In gaseous triethyl amine the C-N-C bond angle is _________ degree.
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2021 Shift 1 - Chemistry - Question 53
The difference between bond orders of CO and NO$$^{\oplus}$$ is $$\frac{x}{2}$$ where x = _________ (Round off to the Nearest Integer)
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2021 Shift 1 - Chemistry - Question 54
For water at 100°C and 1 bar,
$$\Delta_{vap}H - \Delta_{vap}U = \_ \times 10^2$$ J mol$$^{-1}$$
(Round off to the Nearest Integer)
[Use: R = 8.31 J mol$$^{-1}$$ K$$^{-1}$$]
[Assume volume of H$$_2$$O(l) is much smaller than volume of H$$_2$$O(g). Assume H$$_2$$O(g) treated as an ideal gas]
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2021 Shift 1 - Chemistry - Question 55
$$PCl_5 \rightleftharpoons PCl_3 + Cl_2 \quad K_c = 1.844$$
3.0 moles of PCl$$_5$$ is introduced in a 1L closed reaction vessel at 380 K. The number of moles of PCl$$_5$$ at equilibrium is _________ $$\times 10^{-3}$$ (Round off to the Nearest Integer)
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2021 Shift 1 - Chemistry - Question 56
An organic compound is subjected to chlorination to get compound A using 5.0 g of chlorine. When 0.5 g of compound A is reacted with AgNO$$_3$$ [Carius Method], the percentage of chlorine in compound A is when it forms 0.3849 g of AgCl. (Round off to the Nearest Integer)
(Atomic masses of Ag and Cl are 107.87 and 35.5 respectively)
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2021 Shift 1 - Chemistry - Question 57
1.46 g of a biopolymer dissolved in a 100 mL water at 300 K exerted an osmotic pressure of $$2.42 \times 10^{-3}$$ bar.
The molar mass of the biopolymer is _________ $$\times 10^4$$ g mol$$^{-1}$$. (Round off to the Nearest Integer)
[Use: R = 0.083 L bar mol$$^{-1}$$ K$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2021 Shift 1 - Chemistry - Question 58
The conductivity of a weak acid HA of concentration 0.001 mol L$$^{-1}$$ is $$2.0 \times 10^{-5}$$ S cm$$^{-1}$$. If $$\Lambda_m^0(HA) = 190$$ S cm$$^2$$ mol$$^{-1}$$, the ionization constant (K$$_a$$) of HA is equal to _________ $$\times 10^{-6}$$ (Round off to the Nearest Integer)
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2021 Shift 1 - Chemistry - Question 59
CO$$_2$$ gas adsorbs on charcoal following Freundlich adsorption isotherm. For a given amount of charcoal, the mass of CO$$_2$$ adsorbed becomes 64 times when the pressure of CO$$_2$$ is doubled.
The value of n in the Freundlich isotherm equation is _________ $$\times 10^2$$. (Round off to the Nearest Integer)
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2021 Shift 1 - Chemistry - Question 60
The number of geometrical isomers possible in triamminetrinitrocobalt (III) is X and in trioxalatochromate (III) is Y. Then the value of X + Y is _________.
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




