NTA JEE Main 26th July 2022 Shift 2
For the following questions answer them individually
NTA JEE Main 26th July 2022 Shift 2 - Question 41
At $$30°C$$, the half life for the decomposition of $$AB_2$$ is $$200 \text{ s}$$ and is independent of the initial concentration of $$AB_2$$. The time required for $$80\%$$ of the $$AB_2$$ to decompose is (Given: $$\log 2 = 0.30$$; $$\log 3 = 0.48$$)
NTA JEE Main 26th July 2022 Shift 2 - Question 42
Given below are two statements:one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: Finest gold is red in colour, as the size of the particles increases, it appears purple then blue and finally gold.
Reason R: The colour of the colloidal solution depends on the wavelength of light scattered by the dispersed particles.
In the light of the above statements, choose the most appropriate answer from the options given below
NTA JEE Main 26th July 2022 Shift 2 - Question 43
The metal that is not extracted from its sulphide ore is
NTA JEE Main 26th July 2022 Shift 2 - Question 44
The products obtained from a reaction of hydrogen peroxide and acidified potassium permanganate are
NTA JEE Main 26th July 2022 Shift 2 - Question 45
The metal complex that is diamagnetic is (Atomic number: Fe, 26; Cu, 29)
NTA JEE Main 26th July 2022 Shift 2 - Question 46
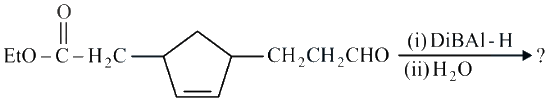
Consider the above reaction and predict the major product.
NTA JEE Main 26th July 2022 Shift 2 - Question 47
Hydrolysis of which compound will give carbolic acid?
NTA JEE Main 26th July 2022 Shift 2 - Question 48
The correct sequential order of the reagents for the given reaction is
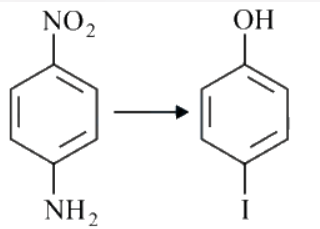
NTA JEE Main 26th July 2022 Shift 2 - Question 49
Vulcanization of rubber is carried out by heating a mixture of
NTA JEE Main 26th July 2022 Shift 2 - Question 50
Animal starch is the other name of


