NTA JEE Main 26th July 2022 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 26th July 2022 Shift 2 - Chemistry - Question 31
Hemoglobin contains $$0.34\%$$ of iron by mass. The number of Fe atoms in $$3.3 \text{ g}$$ of hemoglobin is (Given: Atomic mass of Fe is $$56u$$, $$N_A = 6.022 \times 10^{23} \text{ mol}^{-1}$$)
NTA JEE Main 26th July 2022 Shift 2 - Chemistry - Question 32
The metal that has very low melting point and its periodic position is closer to a metalloid is
NTA JEE Main 26th July 2022 Shift 2 - Chemistry - Question 33
Arrange the following in increasing order of their covalent character.
(A) $$CaF_2$$
(B) $$CaCl_2$$
(C) $$CaBr_2$$
(D) $$CaI_2$$
Choose the correct answer from the options given below.
NTA JEE Main 26th July 2022 Shift 2 - Chemistry - Question 34
Class XII students were asked to prepare one litre of buffer solution of pH $$8.26$$ by their chemistry teacher. The amount of ammonium chloride to be dissolved by the student in $$0.2M$$ ammonia solution to make one litre of the buffer is (Given $$pK_b(NH_3) = 4.74$$; Molar mass of $$NH_3 = 17 \text{ g mol}^{-1}$$. Molar mass of $$NH_4Cl = 53.5 \text{ g mol}^{-1}$$)
NTA JEE Main 26th July 2022 Shift 2 - Chemistry - Question 35
Given below are two statements:one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: Phenolphthalein is a pH dependent indicator, remains colourless in acidic solution and gives pink colour in basic medium.
Reason R: Phenolphthalein is a weak acid. It doesn't dissociate in basic medium.
In the light of the above statements, choose the most appropriate answer from the options given below
NTA JEE Main 26th July 2022 Shift 2 - Chemistry - Question 36
Given below are two statements:one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: LiF is sparingly soluble in water.
Reason R: The ionic radius of $$Li^+$$ ion is smallest among its group members, hence has least hydration enthalpy.
In the light of the above statements, choose the most appropriate answer from the options given below.
NTA JEE Main 26th July 2022 Shift 2 - Chemistry - Question 37
Given below are two statements:one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: Boric acid is a weak acid.
Reason R: Boric acid is not able to release $$H^+$$ ion on its own. It receives $$OH^-$$ ion from water and releases $$H^+$$ ion.
In the light of the above statements, choose the most appropriate answer from the options given below.
NTA JEE Main 26th July 2022 Shift 2 - Chemistry - Question 38
The correct decreasing order of priority of functional groups in naming an organic compound as per IUPAC system of nomenclature is
NTA JEE Main 26th July 2022 Shift 2 - Chemistry - Question 39
Which of the following is not an example of benzenoid compound?
NTA JEE Main 26th July 2022 Shift 2 - Chemistry - Question 40
Match List I with List II

Choose the correct answer from the options given below
NTA JEE Main 26th July 2022 Shift 2 - Chemistry - Question 41
At $$30°C$$, the half life for the decomposition of $$AB_2$$ is $$200 \text{ s}$$ and is independent of the initial concentration of $$AB_2$$. The time required for $$80\%$$ of the $$AB_2$$ to decompose is (Given: $$\log 2 = 0.30$$; $$\log 3 = 0.48$$)
NTA JEE Main 26th July 2022 Shift 2 - Chemistry - Question 42
Given below are two statements:one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: Finest gold is red in colour, as the size of the particles increases, it appears purple then blue and finally gold.
Reason R: The colour of the colloidal solution depends on the wavelength of light scattered by the dispersed particles.
In the light of the above statements, choose the most appropriate answer from the options given below
NTA JEE Main 26th July 2022 Shift 2 - Chemistry - Question 43
The metal that is not extracted from its sulphide ore is
NTA JEE Main 26th July 2022 Shift 2 - Chemistry - Question 44
The products obtained from a reaction of hydrogen peroxide and acidified potassium permanganate are
NTA JEE Main 26th July 2022 Shift 2 - Chemistry - Question 45
The metal complex that is diamagnetic is (Atomic number: Fe, 26; Cu, 29)
NTA JEE Main 26th July 2022 Shift 2 - Chemistry - Question 46
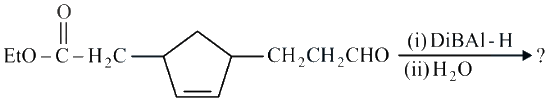
Consider the above reaction and predict the major product.
NTA JEE Main 26th July 2022 Shift 2 - Chemistry - Question 47
Hydrolysis of which compound will give carbolic acid?
NTA JEE Main 26th July 2022 Shift 2 - Chemistry - Question 48
The correct sequential order of the reagents for the given reaction is
NTA JEE Main 26th July 2022 Shift 2 - Chemistry - Question 49
Vulcanization of rubber is carried out by heating a mixture of
NTA JEE Main 26th July 2022 Shift 2 - Chemistry - Question 50
Animal starch is the other name of
NTA JEE Main 26th July 2022 Shift 2 - Chemistry - Question 51
Consider an imaginary ion $$_{22}^{48}X^{3-}$$. The nucleus contains '$$a$$' $$\%$$ more neutrons than the number of electrons in the ion. The value of '$$a$$' is ______.
789
456
123
0.-
Clear All
NTA JEE Main 26th July 2022 Shift 2 - Chemistry - Question 52
A $$10 \text{ g}$$ mixture of hydrogen and helium is contained in a vessel of capacity $$0.0125 \text{ m}^3$$ at $$6 \text{ bar}$$ and $$27°C$$. The mass of helium in the mixture is ______ g. (Given: $$R = 8.3 \text{ J K}^{-1} \text{ mol}^{-1}$$)(Atomic masses of H and He are 1u and 4u, respectively)
789
456
123
0.-
Clear All
NTA JEE Main 26th July 2022 Shift 2 - Chemistry - Question 53
For the reaction $$H_2F_2(g) \rightarrow H_2(g) + F_2(g)$$
$$\Delta U = -59.6 \text{ kJ mol}^{-1}$$ at $$27°C$$
The enthalpy change for the above reaction is $$-$$ ______ $$\text{kJ mol}^{-1}$$ (nearest integer) (Given: $$R = 8.314 \text{ J K}^{-1} \text{ mol}^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 26th July 2022 Shift 2 - Chemistry - Question 54
$$20 \text{ mL}$$ of $$0.02M$$ hypo solution is used for the titration of $$10 \text{ mL}$$ of copper sulphate solution, in the presence of excess of KI using starch as an indicator. The molarity of $$Cu^{2+}$$ is found to be ______ $$\times 10^{-2} M$$ (nearest integer)
Given : $$2Cu^{2+}+4I^{-}\rightarrow Cu_{2}I_{2} + I_{2}I_{2} + 2S_{2}O_{3}^{-2} \rightarrow 2I^{-} + S_{4}O_{6}^{-2}$$
789
456
123
0.-
Clear All
NTA JEE Main 26th July 2022 Shift 2 - Chemistry - Question 55
The elevation in boiling point for 1 molal solution of non-volatile solute A is $$3 \text{ K}$$. The depression in freezing point for 2 molal solution of A in the same solvent is $$6 \text{ K}$$. The ratio of $$K_b$$ and $$K_f$$ i.e., $$K_b/K_f$$ is $$1:X$$. The value of $$X$$ is ______.
789
456
123
0.-
Clear All
NTA JEE Main 26th July 2022 Shift 2 - Chemistry - Question 56
The number of non-ionisable protons present in the product B obtained from the following reaction is ______
$$C_2H_5OH + PCl_3 \rightarrow C_2H_5Cl + A$$
$$A + PCl_3 \rightarrow B$$
789
456
123
0.-
Clear All
NTA JEE Main 26th July 2022 Shift 2 - Chemistry - Question 57
The spin-only magnetic moment value of the compound with strongest oxidizing ability among $$MnF_4$$, $$MnF_3$$ and $$MnF_2$$ is ______ B.M. (nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 26th July 2022 Shift 2 - Chemistry - Question 58
Total number of isomers (including stereoisomers) obtained on monochlorination of methylcyclohexane is ______.
789
456
123
0.-
Clear All
NTA JEE Main 26th July 2022 Shift 2 - Chemistry - Question 59
A $$100 \text{ mL}$$ solution of $$CH_3CH_2MgBr$$ on treatment with methanol produces $$2.24 \text{ mL}$$ of a gas at STP. The weight of gas produced is ______ mg (nearest integer).
789
456
123
0.-
Clear All
NTA JEE Main 26th July 2022 Shift 2 - Chemistry - Question 60
How many of the following drugs is/are example(s) of broad spectrum antibiotic? Ofloxacin, Penicillin G, Terpineol, Salvarsan
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




