NTA JEE Main 26th February 2021 Shift 2
For the following questions answer them individually
NTA JEE Main 26th February 2021 Shift 2 - Question 31
The correct order of electron gain enthalpy is:
NTA JEE Main 26th February 2021 Shift 2 - Question 32
Which pair of oxides is acidic in nature?
NTA JEE Main 26th February 2021 Shift 2 - Question 33
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: In $$TlI_3$$, isomorphous to $$CsI_3$$, the metal is present in +1 oxidation state.
Reason R: Tl metal has fourteen f electrons in its electronic configuration.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 26th February 2021 Shift 2 - Question 34
Match List-I with List-II.
| List-I (Molecule) | List-II (Bond order) |
|---|---|
| (a) $$Ne_2$$ | (i) 1 |
| (b) $$N_2$$ | (ii) 2 |
| (c) $$F_2$$ | (iii) 0 |
| (d) $$O_2$$ | (iv) 3 |
Choose the correct answer from the options given below:
NTA JEE Main 26th February 2021 Shift 2 - Question 35
Calgon is used for water treatment. Which of the following statement is NOT true about Calgon?
NTA JEE Main 26th February 2021 Shift 2 - Question 36
Which of the following forms of hydrogen emits low energy $$\beta^-$$ particles?
NTA JEE Main 26th February 2021 Shift 2 - Question 37
In
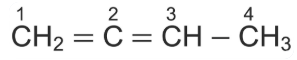
molecule, the hybridization of carbon 1, 2, 3 and 4 respectively, are:
NTA JEE Main 26th February 2021 Shift 2 - Question 38
The nature of charge on resulting colloidal particles when $$FeCl_3$$ is added to excess of hot water is:
NTA JEE Main 26th February 2021 Shift 2 - Question 39
Match List-I with List-II.
| List-I | List-II |
|---|---|
| (a) Sodium Carbonate | (i) Deacon |
| (b) Titanium | (ii) Castner-Kellner |
| (c) Chlorine | (iii) van-Arkel |
| (d) Sodium hydroxide | (iv) Solvay |
Choose the correct answer from the options given below:
NTA JEE Main 26th February 2021 Shift 2 - Question 40
Match List-I with List-II.
| List-I (Ore) | List-II (Metal) |
|---|---|
| (a) Siderite | (i) Cu |
| (b) Calamine | (ii) Ca |
| (c) Malachite | (iii) Fe |
| (d) Cryolite | (iv) Al |
| (v) Zn |
Choose the correct answer from the options given below:


