NTA JEE Main 26th August 2021 Shift 2
For the following questions answer them individually
NTA JEE Main 26th August 2021 Shift 2 - Question 31
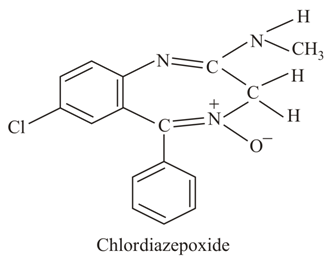
The class of drug to which chlordiazepoxide with above structure belongs is:
NTA JEE Main 26th August 2021 Shift 2 - Question 32
The interaction energy of London forces between two particles is proportional to $$r^x$$, where r is the distance between the particles. The value of x is:
NTA JEE Main 26th August 2021 Shift 2 - Question 33
The bond order and magnetic behaviour of $$O_2^-$$ ion are, respectively:
NTA JEE Main 26th August 2021 Shift 2 - Question 34
Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A) : Heavy water is used for the study of reaction mechanism.
Reason (R): The rate of reaction for the cleavage of O-H bond is slower than that of O-D bond.
Choose the most appropriate answer from the options given below:
NTA JEE Main 26th August 2021 Shift 2 - Question 35
Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A): Barium carbonate is insoluble in water and is highly stable.
Reason (R): The thermal stability of the carbonates increases with increasing cationic size.
Choose the most appropriate answer from the options given below:
NTA JEE Main 26th August 2021 Shift 2 - Question 36
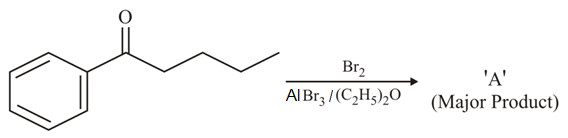
Consider the given reaction, the Product A is:
NTA JEE Main 26th August 2021 Shift 2 - Question 37
Which one of the following compounds is not aromatic?
NTA JEE Main 26th August 2021 Shift 2 - Question 38
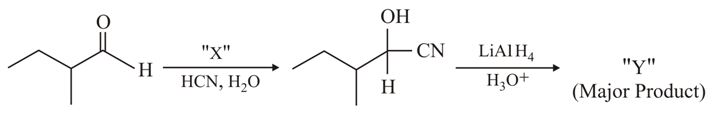
Consider the given reaction, Identify X and Y:
NTA JEE Main 26th August 2021 Shift 2 - Question 39
Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R)
Assertion (A) : Photochemical smog causes cracking of rubber.
Reason (R): Presence of ozone, nitric oxide, acrolein, formaldehyde and peroxyacetyl nitrate in photochemical smog makes it oxidizing.
Choose the most appropriate answer from the options given below:
NTA JEE Main 26th August 2021 Shift 2 - Question 40
The sol given below with negatively charged colloidal particles is:


.webp)
