NTA JEE Main 25th July 2021 Shift 1
For the following questions answer them individually
NTA JEE Main 25th July 2021 Shift 1 - Question 31
The ionic radii of K$$^+$$, Na$$^+$$, Al$$^{3+}$$ and Mg$$^{2+}$$ are in the order:
NTA JEE Main 25th July 2021 Shift 1 - Question 32
At 298.2 K the relationship between enthalpy of bond dissociation (in kJ mol$$^{-1}$$) for hydrogen E$$_H$$ and its isotope, deuterium E$$_D$$, is best described by:
NTA JEE Main 25th July 2021 Shift 1 - Question 33
Given below are two statements:
Statement I : None of the alkaline earth metal hydroxides dissolve in alkali.
Statement II : Solubility of alkaline earth metal hydroxides in water increases down the group.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 25th July 2021 Shift 1 - Question 34
Which one of the following compounds of Group-14 elements is not known?
NTA JEE Main 25th July 2021 Shift 1 - Question 35
Which one among the following resonating structures is not correct?
NTA JEE Main 25th July 2021 Shift 1 - Question 36
An organic compound 'A' C$$_4$$H$$_8$$ on treatment with KMnO$$_4$$/H$$^+$$ yields compound 'B' C$$_3$$H$$_6$$O. Compound 'A' also yields compound 'B' on ozonolysis. Compound 'A' is:
NTA JEE Main 25th July 2021 Shift 1 - Question 37
Which one of the following chemical agent is not being used for dry-cleaning of clothes?
NTA JEE Main 25th July 2021 Shift 1 - Question 38
For the following graphs,
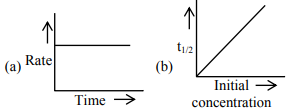
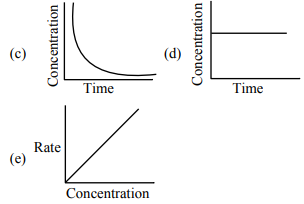
Choose from the options given below, the correct one regarding order of reaction is:
NTA JEE Main 25th July 2021 Shift 1 - Question 39
In the leaching of alumina from bauxite, the ore expected to leach out in the process by reacting with NaOH is:
NTA JEE Main 25th July 2021 Shift 1 - Question 40
The correct order of following 3d metal oxides, according to their oxidation numbers is:
(a) CrO$$_3$$ (b) Fe$$_2$$O$$_3$$ (c) MnO$$_2$$ (d) V$$_2$$O$$_5$$ (e) Cu$$_2$$O


