NTA JEE Main 25th January 2023 Shift 1
For the following questions answer them individually
NTA JEE Main 25th January 2023 Shift 1 - Question 51
The total number of lone pairs of electrons on oxygen atoms of ozone is
789
456
123
0.-
Clear All
NTA JEE Main 25th January 2023 Shift 1 - Question 52
A litre of buffer solution contains 0.1 mole of each of NH$$_3$$ and NH$$_4$$Cl. On the addition of 0.02 mole of HCl by dissolving gaseous HCl, the pH of the solution is found to be _____ $$\times 10^{-3}$$ (Nearest integer)
Given: pK$$_b$$(NH$$_3$$) = 4.745
log 2 = 0.301
log 3 = 0.477
T = 298 K
789
456
123
0.-
Clear All
NTA JEE Main 25th January 2023 Shift 1 - Question 53
The density of a monobasic strong acid (Molar mass 24.2 g mol) is 1.21 kg L. The volume of its solution required for the complete neutralization of 25 mL of 0.24 M NaOH is $$10^{-2}$$ mL (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 25th January 2023 Shift 1 - Question 54
In sulphur estimation. 0.471 g of an organic compound gave 1.4439 g of barium sulphate. The percentage of sulphur in the compound is _____ (Nearest Integer)
(Given: Atomic mass Ba: 137u, S: 32u, O: 16u)
789
456
123
0.-
Clear All
NTA JEE Main 25th January 2023 Shift 1 - Question 55
The osmotic pressure of solutions of PVC in cyclohexanone at 300 K are plotted on the graph. The molar mass of PVC is _____ g mol$$^{-1}$$ (Nearest integer)
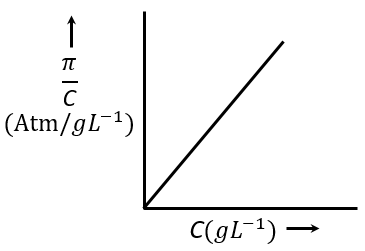
(Given: R = 0.083 L atm K$$^{-1}$$ mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 25th January 2023 Shift 1 - Question 56
Consider the cell Pt(s)|H$$_2$$(s)(1atm)|H$$^+$$(aq, [H$$^+$$] = 1)||Fe$$^{3+}$$(aq), Fe$$^{2+}$$(aq)|Pt(s)
Given: E$$_{Fe^{3+}/Fe^{2+}}^\circ$$ = 0.771 V and E$$_{H^+/\frac{1}{2}H_2}^\circ$$ = 0 V, T = 298 K
If the potential of the cell is 0.712 V the ratio of concentration of Fe$$^{2+}$$ to Fe$$^{3+}$$ is _____ (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 25th January 2023 Shift 1 - Question 57
For the first order reaction A $$\to$$ B the half life is 30 min. The time taken for 75% completion of the reaction is _____ min. (Nearest integer)
Given: log 2 = 0.3010
log 3 = 0.4771
log 5 = 0.6989
789
456
123
0.-
Clear All
NTA JEE Main 25th January 2023 Shift 1 - Question 58
The number of paramagnetic species from the following is
[Ni(CN)$$_4$$]$$^{2-}$$, [Ni(CO)$$_4$$], [NiCl$$_4$$]$$^{2-}$$
[Fe(CN)$$_6$$]$$^{4-}$$, [Cu(NH$$_3$$)$$_4$$]$$^{2+}$$
[Fe(CN)$$_6$$]$$^{3-}$$ and [Fe(H$$_2$$O)$$_6$$]$$^{2+}$$
789
456
123
0.-
Clear All
NTA JEE Main 25th January 2023 Shift 1 - Question 59
How many of the following metal ions have similar value of spin only magnetic moment in gaseous state?
(Given: Atomic number: V, 23; Cr, 24; Fe, 26; Ni, 28)
V$$^{3+}$$, Cr$$^{3+}$$, Fe$$^{2+}$$, Ni$$^{3+}$$
789
456
123
0.-
Clear All
NTA JEE Main 25th January 2023 Shift 1 - Question 60
An athlete is given 100 g of glucose (C$$_6$$H$$_{12}$$O$$_6$$) for energy. This is equivalent to 1800 kJ of energy. The 50% of this energy gained is utilized by the athlete for sports activities at the event. In order to avoid storage of energy, the weight of extra water he would need to perspire is _____ g (Nearest integer) Assume that there is no other way of consuming stored energy.
Given: The enthalpy of evaporation of water is 45 kJ mol$$^{-1}$$
Molar mass of C, H & O are 12.1 and 16 g mol$$^{-1}$$.
789
456
123
0.-
Clear All


