NTA JEE Main 24th February 2021 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 24th February 2021 Shift 2 - Chemistry - Question 31
According to Bohr's atomic theory:
(A) Kinetic energy of electron is $$\propto \frac{Z^2}{n^2}$$.
(B) The product of velocity (v) of electron and principal quantum number (n), $$'vn' \propto Z^2$$.
(C) Frequency of revolution of electron in an orbit is $$\propto \frac{Z^3}{n^3}$$.
(D) Coulombic force of attraction on the electron is $$\propto \frac{Z^3}{n^4}$$.
Choose the most appropriate answer from the options given below:
NTA JEE Main 24th February 2021 Shift 2 - Chemistry - Question 32
The correct set from the following in which both pairs are in correct order of melting point is:
NTA JEE Main 24th February 2021 Shift 2 - Chemistry - Question 33
The correct shape and I - I - I bond angles respectively in $$I_3^-$$ ion are:
NTA JEE Main 24th February 2021 Shift 2 - Chemistry - Question 34
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: Hydrogen is the most abundant element in the Universe, but it is not the most abundant gas in the troposphere.
Reason R: Hydrogen is the lightest element.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Main 24th February 2021 Shift 2 - Chemistry - Question 35
Match List - I with List - II.
| List-I (Salt) | List-II (Flame colour wavelength) |
|---|---|
| (a) LiCl | (i) 455.5 nm |
| (b) NaCl | (ii) 670.8 nm |
| (c) RbCl | (iii) 780.0 nm |
| (d) CsCl | (iv) 589.2 nm |
Choose the correct answer from the options given below:
NTA JEE Main 24th February 2021 Shift 2 - Chemistry - Question 36
Which one of the following compounds is non-aromatic?
NTA JEE Main 24th February 2021 Shift 2 - Chemistry - Question 37
What is the correct sequence of reagents used for converting nitrobenzene into m-dibromobenzene?
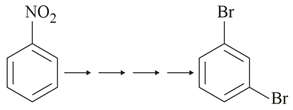
NTA JEE Main 24th February 2021 Shift 2 - Chemistry - Question 38
The correct order of the following compounds showing increasing tendency towards nucleophilic substitution reaction is:
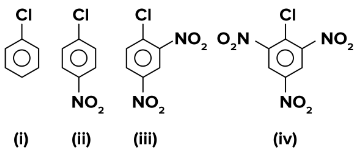
NTA JEE Main 24th February 2021 Shift 2 - Chemistry - Question 39
Given below are two statements:
Statement I: The value of the parameter "Biochemical Oxygen Demand (BOD)" is important for survival of aquatic life.
Statement II: The optimum value of BOD is 6.5 ppm.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 24th February 2021 Shift 2 - Chemistry - Question 40
Most suitable salt which can be used for efficient clotting of blood will be?
NTA JEE Main 24th February 2021 Shift 2 - Chemistry - Question 41
Match List - I with List - II.
| List-I (Metal) | List-II (Ores) |
|---|---|
| (a) Aluminium | (i) Siderite |
| (b) Iron | (ii) Calamine |
| (c) Copper | (iii) Kaolinite |
| (d) Zinc | (iv) Malachite |
Choose the correct answer from the options given below:
NTA JEE Main 24th February 2021 Shift 2 - Chemistry - Question 42
The incorrect statement among the following is:
NTA JEE Main 24th February 2021 Shift 2 - Chemistry - Question 43
What is the correct order of the following elements with respect to their density?
NTA JEE Main 24th February 2021 Shift 2 - Chemistry - Question 44
The calculated magnetic moments (spin only value) for species $$[FeCl_4]^{2-}$$, $$[Co(C_2O_4)_3]^{3-}$$ and $$MnO_4^{2-}$$ respectively are:
NTA JEE Main 24th February 2021 Shift 2 - Chemistry - Question 45
Which of the following reagent is suitable for the preparation of the product in the above reaction?
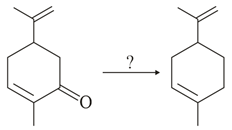
NTA JEE Main 24th February 2021 Shift 2 - Chemistry - Question 46
Which one of the following carbonyl compounds cannot be prepared by addition of water on an alkyne in the presence of $$HgSO_4$$ and $$H_2SO_4$$?
NTA JEE Main 24th February 2021 Shift 2 - Chemistry - Question 47
The diazonium salt of which of the following compounds will form a coloured dye on reaction with $$\beta$$-Naphthol in NaOH?
NTA JEE Main 24th February 2021 Shift 2 - Chemistry - Question 48
Match List - I with List - II.
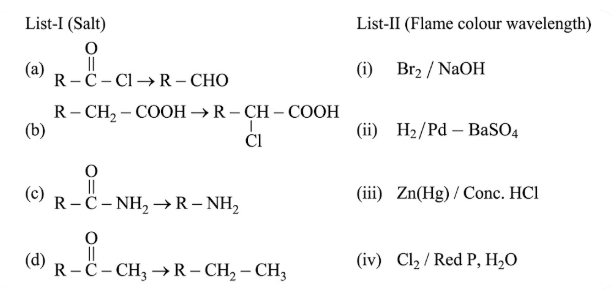
Choose the correct answer from the options given below:
NTA JEE Main 24th February 2021 Shift 2 - Chemistry - Question 49
In polymer Buna-S, 'S' stands for:
NTA JEE Main 24th February 2021 Shift 2 - Chemistry - Question 50
Match List - I with List - II.
| List-I | List-II |
|---|---|
| (a) Valium | (i) Antifertility drug |
| (b) Morphine | (ii) Pernicious anaemia |
| (c) Norethindrone | (iii) Analgesic |
| (d) Vitamin B$$_{12}$$ | (iv) Tranquilizer |
Choose the correct answer from the options given below:
NTA JEE Main 24th February 2021 Shift 2 - Chemistry - Question 51
The formula of a gaseous hydrocarbon which requires 6 times of its own volume of $$O_2$$ for complete oxidation and produces 4 times its own volume of $$CO_2$$ is $$C_xH_y$$. The value of y is ______.
789
456
123
0.-
Clear All
NTA JEE Main 24th February 2021 Shift 2 - Chemistry - Question 52
1.86 g of aniline completely reacts to form acetanilide. 10% of the product is lost during purification. Amount of acetanilide obtained after purification (in g) is ______ $$\times 10^{-2}$$.
789
456
123
0.-
Clear All
NTA JEE Main 24th February 2021 Shift 2 - Chemistry - Question 53
The volume occupied by 4.75 g of acetylene gas at 50°C and 740 mm Hg pressure is ______ L.
(Rounded off to the nearest integer)
[Given R = 0.0826 L atm K$$^{-1}$$ mol$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 24th February 2021 Shift 2 - Chemistry - Question 54
Assuming ideal behaviour, the magnitude of log K for the following reaction at 25°C is $$x \times 10^{-1}$$. The value of x is ______. (Integer answer)
$$3HC \equiv CH_{(g)} \rightleftharpoons C_6H_{6(l)}$$
[Given: $$\Delta_f G^{\circ}(HC \equiv CH) = -2.04 \times 10^5$$ J mol$$^{-1}$$; $$\Delta_f G^{\circ}(C_6H_6) = -1.24 \times 10^5$$ J mol$$^{-1}$$; R = 8.314 J K$$^{-1}$$ mol$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 24th February 2021 Shift 2 - Chemistry - Question 55
The solubility product of $$PbI_2$$ is $$8.0 \times 10^{-9}$$. The solubility of lead iodide in 0.1 molar solution of lead nitrate is $$x \times 10^{-6}$$ mol/L. The value of x is ______ (Rounded off to the nearest integer)
[Given $$\sqrt{2} = 1.41$$]
789
456
123
0.-
Clear All
NTA JEE Main 24th February 2021 Shift 2 - Chemistry - Question 56
$$C_6H_6$$ freezes at 5.5°C. The temperature at which a solution of 10 g of $$C_4H_{10}$$ in 200 g of $$C_6H_6$$ freeze is ______ °C. (nearest integer value), (The molal freezing point depression constant of $$C_6H_6$$ is 5.12°C/m.)
789
456
123
0.-
Clear All
NTA JEE Main 24th February 2021 Shift 2 - Chemistry - Question 57
The magnitude of the change in oxidising power of the $$MnO_4^- / Mn^{2+}$$ couple is $$x \times 10^{-4}$$ V if the $$H^+$$ concentration is decreased from 1 M to $$10^{-4}$$ M at 25°C. (Assume concentration of $$MnO_4^-$$ and $$Mn^{2+}$$ to be same on change in $$H^+$$ concentration). The value of x is ______. (Rounded off to the nearest integer)
[Given: $$\frac{2.303RT}{F} = 0.059$$]
789
456
123
0.-
Clear All
NTA JEE Main 24th February 2021 Shift 2 - Chemistry - Question 58
Sucrose hydrolyses in acid solution into glucose and fructose following first order rate law with a half-life of 3.33 h at 25°C. After 9 h, the fraction of sucrose remaining is f. The value of $$\log_{10}\left(\frac{1}{f}\right)$$ is ______ $$\times 10^{-2}$$.
(Rounded off to the nearest integer)
[Assume: ln10 = 2.303, ln 2 = 0.693]
789
456
123
0.-
Clear All
NTA JEE Main 24th February 2021 Shift 2 - Chemistry - Question 59
Among the following allotropic forms of sulphur, the number of allotropic forms, which will show paramagnetism is ______.
(A) $$\alpha$$-sulphur
(B) $$\beta$$-sulphur
(C) $$S_2$$-form
789
456
123
0.-
Clear All
NTA JEE Main 24th February 2021 Shift 2 - Chemistry - Question 60
The total number of amines among the following which can be synthesized by Gabriel synthesis is ______.
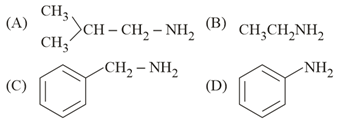
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




