NTA JEE Main 2025 April 4th Shift 2
For the following questions answer them individually
NTA JEE Main 2025 April 4th Shift 2 - Question 61
The correct order of $$[FeF_6]^{3-}$$, $$[CoF_6]^{3-}$$, $$[Ni(CO)_4]$$ and $$[Ni(CN)_4]^{2-}$$ complex species based on the number of unpaired electrons present is :
NTA JEE Main 2025 April 4th Shift 2 - Question 62
Consider the given data :
(a) $$HCl(g) + 10H_2O(l) \rightarrow HCl \cdot 10H_2O$$, $$\Delta H = -69.01$$ kJ mol$$^{-1}$$
(b) $$HCl(g) + 40H_2O(l) \rightarrow HCl \cdot 40H_2O$$, $$\Delta H = -72.79$$ kJ mol$$^{-1}$$
Choose the correct statement :
NTA JEE Main 2025 April 4th Shift 2 - Question 63
Consider the ground state of chromium atom (Z = 24). How many electrons are with Azimuthal quantum number $$l = 1$$ and $$l = 2$$ respectively?
NTA JEE Main 2025 April 4th Shift 2 - Question 64
Given below are two statements :
Statement (I) : The first ionisation enthalpy of group 14 elements is higher than the corresponding elements of group 13.
Statement (II) : Melting points and boiling points of group 13 elements are in general much higher than those the corresponding elements of group 14.
In the light of the above statements, choose the most appropriate answer from the options given below :
NTA JEE Main 2025 April 4th Shift 2 - Question 65
Consider the following plots of log of rate constant k (log k) vs $$\frac{1}{T}$$ for three different reactions. The correct order of activation energies of these reactions is :
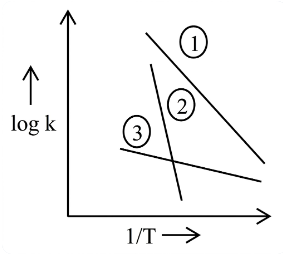
NTA JEE Main 2025 April 4th Shift 2 - Question 66
'X' is the number of electrons in $$t_{2g}$$ orbitals of the most stable complex ion among $$[Fe(NH_3)_6]^{3+}$$, $$[Fe(Cl)_6]^{3-}$$, $$[Fe(C_2O_4)_3]^{3-}$$ and $$[Fe(H_2O)_6]^{3+}$$. The nature of oxide of vanadium of the type $$V_2O_x$$ is:
NTA JEE Main 2025 April 4th Shift 2 - Question 67
The elements of Group 13 with highest and lowest first ionisation enthalpies are respectively:
NTA JEE Main 2025 April 4th Shift 2 - Question 68
Consider the following molecule (X). The structure of X is
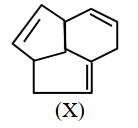
NTA JEE Main 2025 April 4th Shift 2 - Question 69
Given below are two statements:
Statement (I) : for

, all three possible structures may be drawn.
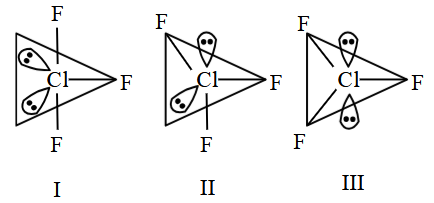
Statement (II) : Structure III is most stable, as the orbitals having the lone pairs are axial, where the $$lp-bp$$ repulsion is minimum.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 2025 April 4th Shift 2 - Question 70
Half life of zero order reaction $$A \rightarrow$$ product is 1 hour, when initial concentration of reaction is 2.0 mol L$$^{-1}$$. The time required to decrease concentration of A from 0.50 to 0.25 mol L$$^{-1}$$ is:


