NTA JEE Main 2025 April 4th Shift 1
For the following questions answer them individually
NTA JEE Main 2025 April 4th Shift 1 - Question 61
On charging the lead storage battery, the oxidation state of lead changes from $$x_1$$ to $$y_1$$ at the anode and from $$x_2$$ to $$y_2$$ at the cathode. The values of $$x_1, y_1, x_2, y_2$$ are respectively:
NTA JEE Main 2025 April 4th Shift 1 - Question 62
Given below are two statements:
Statement I: Nitrogen forms oxides with +1 to +5 oxidation states due to the formation of $$p\pi - p\pi$$ bond with oxygen.
Statement II: Nitrogen does not form halides with +5 oxidation state due to the absence of d-orbital in it.
In the light of given statements, choose the correct answer from the options given below.
NTA JEE Main 2025 April 4th Shift 1 - Question 63
Benzene is treated with oleum to produce compound (X) which when further heated with molten sodium hydroxide followed by acidification produces compound (Y). The compound Y is treated with zinc metal to produce compound (Z). Identify the structure of compound (Z) from the following option.
NTA JEE Main 2025 April 4th Shift 1 - Question 64
Identify the pair of reactants that upon reaction, with elimination of HCl will give rise to the dipeptide Gly-Ala.
NTA JEE Main 2025 April 4th Shift 1 - Question 65
Given below are the pairs of group 13 elements showing their relation in terms of atomic radius. $$(B \lt Al), (Al \lt Ga), (Ga \lt In)$$ and $$(In \lt Tl)$$. Identify the elements present in the incorrect pair and in that pair find out the element (X) that has higher ionic radius ($$M^{3+}$$) than the other one. The atomic number of the element (X) is
NTA JEE Main 2025 April 4th Shift 1 - Question 66
An organic compound (X) with molecular formula $$C_3H_6O$$ is not readily oxidised. On reduction it gives $$(C_3H_8O)(Y)$$ which reacts with HBr to give a bromide (Z) which is converted to Grignard reagent. This Grignard reagent on reaction with (X) followed by hydrolysis gives 2,3-dimethylbutan-2-ol. Compounds (X), (Y) and (Z) respectively are:
NTA JEE Main 2025 April 4th Shift 1 - Question 67
Predict the major product of the following reaction sequence:
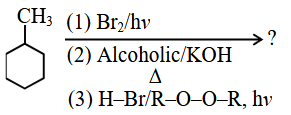
NTA JEE Main 2025 April 4th Shift 1 - Question 68
Given below are two statements:
Statement I: The dipole moment of $$\overset{4}{\mathrm{CH_3}} - \overset{3}{\mathrm{CH}} = \overset{2}{\mathrm{CH}} - \overset{1}{\mathrm{CH}}=O$$ is greater than $$\overset{4}{\mathrm{CH_3}} - \overset{3}{\mathrm{CH_2}} - \overset{2}{\mathrm{CH_2}} - \overset{1}{\mathrm{CH}}=O$$.
Statement II: $$C_1-C_2$$ bond length of $$\underset{4}{\mathrm{CH_3}} - \underset{3}{\mathrm{CH}} = \underset{2}{\mathrm{CH}} - \underset{1}{\mathrm{CH}}=O$$ is greater than $$C_1-C_2$$ bond length of $$\underset{4}{\mathrm{CH_3}} - \underset{3}{\mathrm{CH_2}} - \underset{2}{\mathrm{CH_2}} - \underset{1}{\mathrm{CH}}=O$$.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Main 2025 April 4th Shift 1 - Question 69
Pair of transition metal ions having the same number of unpaired electrons is:
NTA JEE Main 2025 April 4th Shift 1 - Question 70
Which one of the following about an electron occupying the 1s orbital in a hydrogen atom is incorrect? (Bohr's radius is represented by $$a_0$$)


