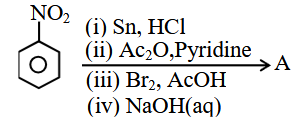NTA JEE Main 2025 April 4th Shift 1
For the following questions answer them individually
NTA JEE Main 2025 April 4th Shift 1 - Question 51
XY is the membrane/partition between two chambers 1 and 2 containing sugar solutions of concentration $$c_1$$ and $$c_2$$ ($$c_1 \gt c_2$$) mol $$L^{-1}$$. For the reverse osmosis to take place identify the correct condition. (Here $$p_1$$ and $$p_2$$ are pressures applied on chamber 1 and 2)
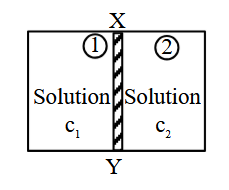
(A) Membrane/Partition ; Cellophane, $$p_{1} > \pi$$
(B) Membrane/Partition ; Porous.$$p_{2} > \pi$$
(C) Membrane/Partition ; Parchment paper, $$p_{1} > \pi$$
(D) Membrane/Partition : Cellophane, $$p_{2} > \pi$$
Choose the correct answer from the options given below :
NTA JEE Main 2025 April 4th Shift 1 - Question 52
Let us consider a reversible reaction at temperature, T. In this reaction, both $$\Delta H$$ and $$\Delta S$$ were observed to have positive values. If the equilibrium temperature is $$T_e$$, then the reaction becomes spontaneous at:
NTA JEE Main 2025 April 4th Shift 1 - Question 53
Which of the following molecules show/s paramagnetic behavior?
(A) $$O_2$$ (B) $$N_2$$ (C) $$F_2$$ (D) $$S_2$$ (E) $$Cl_2$$
Choose the correct answer:
NTA JEE Main 2025 April 4th Shift 1 - Question 54
Aldol condensation is a popular and classical method to prepare $$\alpha,\beta$$-unsaturated carbonyl compounds. This reaction can be both intermolecular and intramolecular. Predict which one of the following is not a product of intramolecular aldol condensation?
NTA JEE Main 2025 April 4th Shift 1 - Question 55
One mole of an ideal gas expands isothermally and reversibly from 10 $$dm^3$$ to 20 $$dm^3$$ at 300 K. $$\Delta U$$, q and work done in the process respectively are: (Given: $$R = 8.3 J K^{-1} mol^{-1}$$, $$\ln 10 = 2.3$$, $$\log 2 = 0.30$$, $$\log 3 = 0.48$$)
NTA JEE Main 2025 April 4th Shift 1 - Question 56
Which one of the following complexes will have $$\Delta_0 = 0$$ and $$\mu = 5.96$$ B.M.?
NTA JEE Main 2025 April 4th Shift 1 - Question 57
For $$A_2 + B_2 \rightleftharpoons 2AB$$. $$E_a$$ for forward and backward reaction are 180 and 200 kJ mol$$^{-1}$$ respectively. If catalyst lowers $$E_a$$ for both reaction by 100 kJ mol$$^{-1}$$. Which of the following statement is correct?
NTA JEE Main 2025 April 4th Shift 1 - Question 58
Rate law for a reaction between A and B is given by $$R = k[A]^n[B]^m$$. If concentration of A is doubled and concentration of B is halved from their initial value, the ratio of new rate of reaction to the initial rate of reaction $$\left(\dfrac{r_2}{r_1}\right)$$ is
NTA JEE Main 2025 April 4th Shift 1 - Question 59
Number of stereoisomers possible for the complexes, $$[CrCl_3(py)_3]$$ and $$[CrCl_2(ox)_2]^{3-}$$ are respectively (py = pyridine, ox = oxalate)
NTA JEE Main 2025 April 4th Shift 1 - Question 60
The major product (A) formed in the following reaction sequence is:
Nitrobenzene $$\xrightarrow{(i) Sn, HCl}$$ $$\xrightarrow{(ii) Ac_2O, Pyridine}$$ $$\xrightarrow{(iii) Br_2, AcOH}$$ $$\xrightarrow{(iv) NaOH(aq)}$$ A