NTA JEE Main 2025 April 3rd Shift 2
For the following questions answer them individually
NTA JEE Main 2025 April 3rd Shift 2 - Question 51
40 mL of a mixture of $$CH_3COOH$$ and HCl (aqueous solution) is titrated against 0.1 M NaOH conductometrically. Which of the following statement is correct?
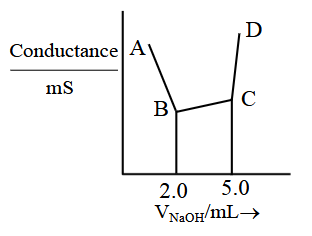
NTA JEE Main 2025 April 3rd Shift 2 - Question 52
10 mL of 2 M NaOH solution is added to 20 mL of 1 M HCl solution kept in a beaker. Now, 10 mL of this mixture is poured into a volumetric flask of 100 mL containing 2 moles of HCl and made the volume upto the mark with distilled water. The solution in this flask is:
NTA JEE Main 2025 April 3rd Shift 2 - Question 53
Fat soluble vitamins are:
A. Vitamin $$B_1$$
B. Vitamin C
C. Vitamin E
D. Vitamin $$B_{12}$$
E. Vitamin K
Choose the correct answer from the options given below:
NTA JEE Main 2025 April 3rd Shift 2 - Question 54
Match the LIST-I with LIST-II.
| LIST-I (Family) | LIST-II (Symbol) | ||
|---|---|---|---|
| A. | Pnicogen (group 15) | I. | Ts |
| B. | Chalcogen | II. | Og |
| C. | Halogen | III. | Lv |
| D. | Noble gas | IV. | Mc |
Choose the correct answer from the options given below :
NTA JEE Main 2025 April 3rd Shift 2 - Question 55
For electron in '2s' and '2p' orbitals, the orbital angular momentum values, respectively are:
NTA JEE Main 2025 April 3rd Shift 2 - Question 56
Compounds that should not be used as primary standards in titrimetric analysis are:
A. $$Na_2Cr_2O_7$$
B. Oxalic acid
C. NaOH
D. $$FeSO_4 \cdot 6H_2O$$
E. Sodium tetraborate
Choose the most appropriate answer from the options given below:
NTA JEE Main 2025 April 3rd Shift 2 - Question 57
The major product (P) in the following reaction is:
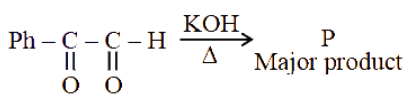
NTA JEE Main 2025 April 3rd Shift 2 - Question 58
In the following series of reactions identify the major products A & B respectively.
Bromobenzene $$\xrightarrow{SO_3, H_2SO_4}$$ A (Major product) $$\xrightarrow{Br_2, Fe}$$ B (Major product)
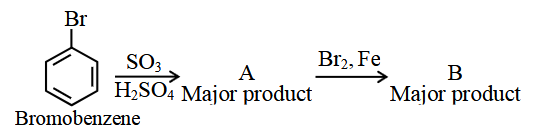
NTA JEE Main 2025 April 3rd Shift 2 - Question 59
The standard cell potential ($$E^\ominus_{\text{cell}}$$) of a fuel cell based on the oxidation of methanol in air that has been used to power television relay station is measured as 1.21 V. The standard half cell reduction potential for $$O_2$$ ($$E^\circ_{O_2/H_2O}$$) is 1.229 V. Choose the correct statement:
NTA JEE Main 2025 April 3rd Shift 2 - Question 60
Identify the diamagnetic octahedral complex ions from below:
A. $$[Mn(CN)_6]^{3-}$$
B. $$[Co(NH_3)_6]^{3+}$$
C. $$[Fe(CN)_6]^{4-}$$
D. $$[Co(H_2O)_3F_3]$$
Choose the correct answer from the options given below:


