NTA JEE Main 2025 April 3rd Shift 2
For the following questions answer them individually
NTA JEE Main 2025 April 3rd Shift 2 - Question 71
X g of nitrobenzene on nitration gave 4.2 g of m-dinitrobenzene. X = ________ g. (nearest integer)
[Given: molar mass (in g mol$$^{-1}$$) C: 12, H: 1, O: 16, N: 14]
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 3rd Shift 2 - Question 72
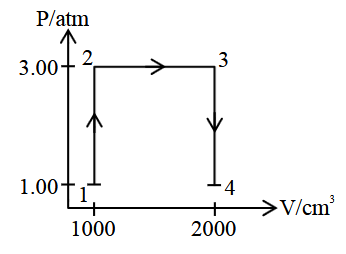
A perfect gas (0.1 mol) having $$\bar{C}_v = 1.50 R$$ (independent of temperature) undergoes the transformation from point 1 to point 4 as shown in the P-V diagram. If each step is reversible, the total work done (w) while going from point 1 to point 4 is $$(-) \text{____}$$ J (nearest integer).
[Given: $$R = 0.082$$ L atm $$K^{-1} mol^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 3rd Shift 2 - Question 73
A sample of n-octane (1.14 g) was completely burnt in excess of oxygen in a bomb calorimeter, whose heat capacity is 5 kJ $$K^{-1}$$. As a result of combustion reaction, the temperature of the calorimeter is increased by 5 K. The magnitude of the heat of combustion of octane at constant volume is ________ kJ mol$$^{-1}$$ (nearest integer).
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 3rd Shift 2 - Question 74
Among Sc, Mn, Co and Cu, identify the element with highest enthalpy of atomisation. The spin only magnetic moment value of that element in its +2 oxidation state is ________ BM (in nearest integer).
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 3rd Shift 2 - Question 75
The total number of structural isomers possible for the substituted benzene derivatives with the molecular formula $$C_9H_{12}$$ is ________.
789
456
123
0.-
Clear All


