NTA JEE Main 2025 April 3rd Shift 1
For the following questions answer them individually
NTA JEE Main 2025 April 3rd Shift 1 - Question 61
Number of molecules from below which cannot give iodoform reaction is :
Ethanol, Isopropyl alcohol, Bromoacetone, 2-Butanol, 2-Butanone, Butanal, 2-Pentanone, 3-Pentanone, Pentanal and 3-Pentanol
NTA JEE Main 2025 April 3rd Shift 1 - Question 62
Identify [A], [B], and [C], respectively in the following reaction sequence :
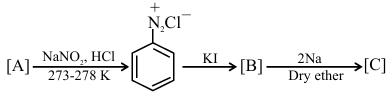
NTA JEE Main 2025 April 3rd Shift 1 - Question 63
In the following reactions, which one is NOT correct?
NTA JEE Main 2025 April 3rd Shift 1 - Question 64
The correct order of the complexes $$[Co(NH_3)_4(H_2O)]^{3+}$$ (A), $$[Co(NH_3)_6]^{3+}$$ (B), $$[Co(CN)_6]^{3-}$$ (C) and $$[CoCl(NH_3)_5]^{2+}$$ (D) in terms of wavelength of light absorbed is :
NTA JEE Main 2025 April 3rd Shift 1 - Question 65
2 moles each of ethylene glycol and glucose are dissolved in 500 g of water. The boiling point of the resulting solution is :
(Given : Ebullioscopic constant of water = 0.52 K kg mol$$^{-1}$$)
NTA JEE Main 2025 April 3rd Shift 1 - Question 66
Which compound would give 3-methyl-6-oxoheptanal upon ozonolysis ?
NTA JEE Main 2025 April 3rd Shift 1 - Question 67
Match the LIST-I with LIST-II.
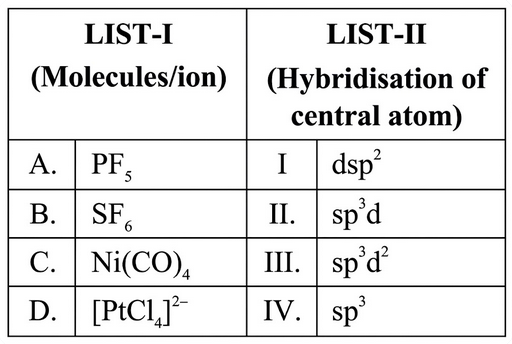
Choose the correct answer from the options given below :
NTA JEE Main 2025 April 3rd Shift 1 - Question 68
The least acidic compound, among the following is:
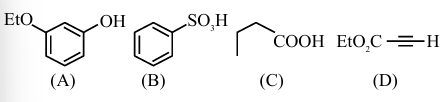
NTA JEE Main 2025 April 3rd Shift 1 - Question 69
Correct order of limiting molar conductivity for cations in water at 298 K is :
NTA JEE Main 2025 April 3rd Shift 1 - Question 70
During estimation of nitrogen by Dumas' method of compound X (0.42 g) :
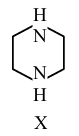
_________ mL of $$N_2$$ gas will be liberated at STP. (nearest integer)
(Given molar mass in g mol$$^{-1}$$ : C : 12, H : 1, N : 14)
789
456
123
0.-
Clear All


