NTA JEE Main 2025 April 3rd Shift 1
For the following questions answer them individually
NTA JEE Main 2025 April 3rd Shift 1 - Question 51
Which of the following postulate of Bohr's model of hydrogen atom is not in agreement with quantum mechanical model of an atom ?
NTA JEE Main 2025 April 3rd Shift 1 - Question 52
Given below are two statements :
Statement I : The N-N single bond is weaker and longer than that of P-P single bond
Statement II : Compounds of group 15 elements in +3 oxidation states readily undergo disproportionation reactions.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Main 2025 April 3rd Shift 1 - Question 53
Given below are two statements
Statement I : A catalyst cannot alter the equilibrium constant $$(K_c)$$ of the reaction, temperature remaining constant
Statement II : A homogeneous catalyst can change the equilibrium composition of a system temperature remaining constant
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Main 2025 April 3rd Shift 1 - Question 54
The metal ions that have the calculated spin only magnetic moment value of 4.9 B.M. are
A. $$Cr^{2+}$$
B. $$Fe^{2+}$$
C. $$Fe^{3+}$$
D. $$Co^{2+}$$
E. $$Mn^{3+}$$
Choose the correct answer from the options given below
NTA JEE Main 2025 April 3rd Shift 1 - Question 55
In a reaction $$A + B \to C$$, initial concentrations of A and B are related as $$[A]_0 = 8[B]_0$$. The half lives of A and B are 10 min and 40 min respectively. If they start to disappear at the same time, both following first order kinetics, after how much time will the concentration of both the reactants be same?
NTA JEE Main 2025 April 3rd Shift 1 - Question 56
Which of the following is the correct structure of L-fructose ?
NTA JEE Main 2025 April 3rd Shift 1 - Question 57
Identify the correct statements from the following
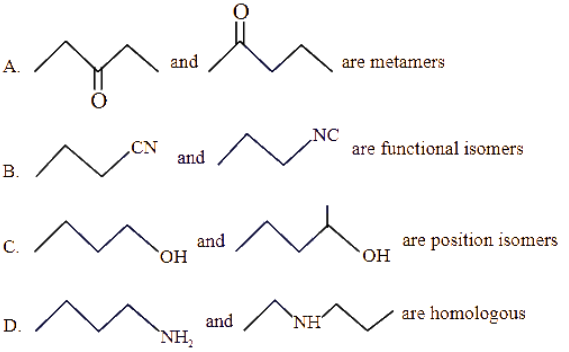
Choose the correct answer from the options given below
NTA JEE Main 2025 April 3rd Shift 1 - Question 58
Among $$10^{-9}$$ g (each) of the following elements, which one will have the highest number of atoms?
Element : Pb, Po, Pr and Pt
NTA JEE Main 2025 April 3rd Shift 1 - Question 59
Which of the following statements are correct?
A. The process of the addition of an electron to a neutral gaseous atom is always exothermic
B. The process of removing an electron from an isolated gaseous atom is always endothermic
C. The 1st ionization energy of the boron is less than that of the beryllium
D. The electronegativity of C is 2.5 in $$CH_4$$ and $$CCl_4$$
E. Li is the most electropositive among elements of group I
Choose the correct answer from the options given below
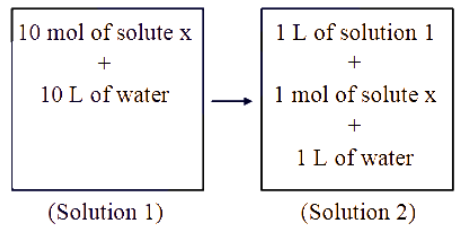
NTA JEE Main 2025 April 3rd Shift 1 - Question 60
Which of the following properties will change when system containing solution 1 will become solution 2 ?