NTA JEE Main 20th July 2021 Shift 1
For the following questions answer them individually
NTA JEE Main 20th July 2021 Shift 1 - Question 51
The Azimuthal quantum number for the valence electrons of Ga$$^+$$ ion is (Atomic number of Ga = 31)
789
456
123
0.-
Clear All
NTA JEE Main 20th July 2021 Shift 1 - Question 52
The number of lone pairs of electrons on the central I atom in I$$_3^-$$ is ___.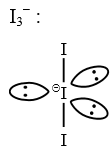
789
456
123
0.-
Clear All
NTA JEE Main 20th July 2021 Shift 1 - Question 53
An average person needs about 10000 kJ energy per day. The amount of glucose (molar mass = 180.0 g mol$$^{-1}$$) needed to meet this energy requirement is ___ g.
(Use : $$\Delta_C$$H (glucose) = -2700 kJ mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 20th July 2021 Shift 1 - Question 54
$$2$$ SO$$_2$$(g) + O$$_2$$(g) $$\rightleftharpoons$$ 2 SO$$_3$$(g)
In an equilibrium mixture, the partial pressures are
P$$_{SO_3}$$ = 43 kPa; P$$_{O_2}$$ = 530 Pa and
P$$_{SO_2}$$ = 45 kPa. The equilibrium constant K$$_P$$ = ___ $$\times 10^{-2}$$. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 20th July 2021 Shift 1 - Question 55
250 mL of 0.5M NaOH was added to 500 mL of 1M HCl. The number of unreacted HCl molecules in the solution is p$$\times 10^{21}$$. Find out p.
(Nearest integer) (N$$_A$$ = 6.022 $$\times 10^{23}$$)
789
456
123
0.-
Clear All
NTA JEE Main 20th July 2021 Shift 1 - Question 56
At 20$$^\circ$$C, the vapour pressure of benzene is 70 torr and that of methyl benzene is 20 torr. The mole fraction of benzene in the vapour phase at 20$$^\circ$$C above an equimolar mixture of benzene and methyl benzene is ___ $$\times 10^{-2}$$. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 20th July 2021 Shift 1 - Question 57
The inactivation rate of a viral preparation is proportional to the amount of virus. In the first minute after preparation, 10% of the virus is inactivated. The rate constant for viral inactivation is ___ $$\times 10^{-3}$$ min$$^{-1}$$.
(Nearest integer) [Use : ln 10 = 2.303; $$\log_{10} 3 = 0.477$$ property of logarithm: $$\log x^y = y \log x$$]
789
456
123
0.-
Clear All
NTA JEE Main 20th July 2021 Shift 1 - Question 58
The spin-only magnetic moment value for the complex [Co(CN)$$_6$$]$$^{4-}$$ is ___ BM. (nearest integer value)
[At. no. of Co = 27]
789
456
123
0.-
Clear All
NTA JEE Main 20th July 2021 Shift 1 - Question 59
To synthesise 1 mole of 2-methylpropan-2-ol from Ethylethanoate equivalents of CH$$_3$$MgBr reagent will be required. (Integer value)
789
456
123
0.-
Clear All
NTA JEE Main 20th July 2021 Shift 1 - Question 60
The number of nitrogen atoms in a semicarbazone molecule of acetone is ___.
789
456
123
0.-
Clear All


