NTA JEE Main 20th July 2021 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 20th July 2021 Shift 1 - Chemistry - Question 31
An inorganic Compound 'X' on treatment with concentrated H$$_2$$SO$$_4$$ produces brown fumes and gives dark brown ring with FeSO$$_4$$ in presence of concentrated H$$_2$$SO$$_4$$. Also Compound 'X' gives precipitate 'Y', when its solution in dilute HCl is treated with H$$_2$$S gas. The precipitate 'Y' on treatment with concentrated HNO$$_3$$ followed by excess of NH$$_4$$OH further gives deep blue coloured solution, Compound 'X' is:
NTA JEE Main 20th July 2021 Shift 1 - Chemistry - Question 32
The set in which compounds have different nature is:
NTA JEE Main 20th July 2021 Shift 1 - Chemistry - Question 33
The species given below that does NOT show disproportionation reaction is:
NTA JEE Main 20th July 2021 Shift 1 - Chemistry - Question 34
Given below are two statements : One is labelled as Assertion A and other is labelled as Reason R.
Assertion A : The dihedral angles in H$$_2$$O$$_2$$ in gaseous phase is 90.2$$^\circ$$ and in solid phase is 111.5$$^\circ$$.
Reason R : The change in dihedral angle in solid and gaseous phase is due to the difference in the intermolecular forces.
Choose the most appropriate answer from the options given below for A and R.
NTA JEE Main 20th July 2021 Shift 1 - Chemistry - Question 35
A s-block element (M) reacts with oxygen to form an oxide of the formula MO$$_2$$. The oxide is pale yellow in colour and paramagnetic. The element (M) is:
NTA JEE Main 20th July 2021 Shift 1 - Chemistry - Question 36
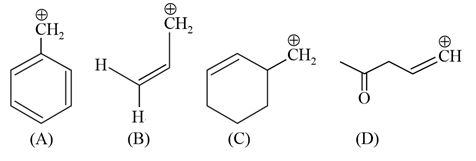
Among the given species the Resonance stabilised carbocations are:
NTA JEE Main 20th July 2021 Shift 1 - Chemistry - Question 37
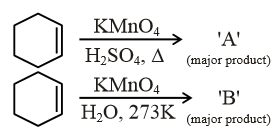
For above chemical reactions, identify the correct statement from the following:
NTA JEE Main 20th July 2021 Shift 1 - Chemistry - Question 38
Green chemistry in day-to-day life is in the use of:
NTA JEE Main 20th July 2021 Shift 1 - Chemistry - Question 39
Given below are two statements. One is labelled as Assertion A and the other is labelled as Reason R.
Assertion A : Sharp glass edge becomes smooth on heating it up to its melting point.
Reason R : The viscosity of glass decreases on melting.
Choose the most appropriate answer from the options given below.
NTA JEE Main 20th July 2021 Shift 1 - Chemistry - Question 40
The conditions given below are in the context of observing Tyndall effect in colloidal solutions:
(A) The diameter of the colloidal particles is comparable to the wavelength of light used.
(B) The diameter of the colloidal particles is much smaller than the wavelength of light used.
(C) The diameter of the colloidal particles is much larger than the wavelength of light used.
(D) The refractive indices of the dispersed phase and the dispersion medium are comparable.
(E) The dispersed phase has a very different refractive index from the dispersion medium. Choose the most appropriate conditions from the options given below:
NTA JEE Main 20th July 2021 Shift 1 - Chemistry - Question 41
The metal that can be purified economically by fractional distillation method is:
NTA JEE Main 20th July 2021 Shift 1 - Chemistry - Question 42
Chemical nature of the nitrogen oxide compound obtained from a reaction of concentrated nitric acid and P$$_4$$O$$_{10}$$ (in 4 : 1 ratio) is:
NTA JEE Main 20th July 2021 Shift 1 - Chemistry - Question 43
According to the valence bond theory the hybridization of central metal atom is dsp$$^2$$ for which one of the following compounds?
NTA JEE Main 20th July 2021 Shift 1 - Chemistry - Question 44
The correct order of intensity of colors of the compounds is:
NTA JEE Main 20th July 2021 Shift 1 - Chemistry - Question 45
In the given reaction 3-Bromo-2, 2-dimethyl butane $$\xrightarrow{C_2H_5OH}$$ 'A' (Major Product). Product A is:
NTA JEE Main 20th July 2021 Shift 1 - Chemistry - Question 46
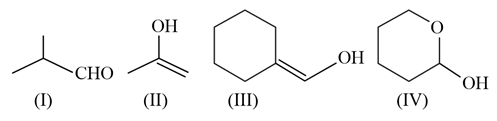
Which among the above compound/s does/do not form Silver mirror when treated with Tollen's reagent?
NTA JEE Main 20th July 2021 Shift 1 - Chemistry - Question 47
Compound A is converted to B on reaction with CHCl$$_3$$ and KOH. The compound B is toxic and can be decomposed by C. A, B and C respectively are:
NTA JEE Main 20th July 2021 Shift 1 - Chemistry - Question 48
Orlon fibres are made up of:
NTA JEE Main 20th July 2021 Shift 1 - Chemistry - Question 49
The correct structure of Ruhemann's Purple, the compound formed in the reaction of Ninhydrin with proteins is:
NTA JEE Main 20th July 2021 Shift 1 - Chemistry - Question 50
Identify the incorrect statement from the following.
NTA JEE Main 20th July 2021 Shift 1 - Chemistry - Question 51
The Azimuthal quantum number for the valence electrons of Ga$$^+$$ ion is (Atomic number of Ga = 31)
789
456
123
0.-
Clear All
NTA JEE Main 20th July 2021 Shift 1 - Chemistry - Question 52
The number of lone pairs of electrons on the central I atom in I$$_3^-$$ is ___.
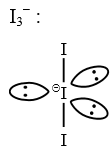
789
456
123
0.-
Clear All
NTA JEE Main 20th July 2021 Shift 1 - Chemistry - Question 53
An average person needs about 10000 kJ energy per day. The amount of glucose (molar mass = 180.0 g mol$$^{-1}$$) needed to meet this energy requirement is ___ g.
(Use : $$\Delta_C$$H (glucose) = -2700 kJ mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 20th July 2021 Shift 1 - Chemistry - Question 54
$$2$$ SO$$_2$$(g) + O$$_2$$(g) $$\rightleftharpoons$$ 2 SO$$_3$$(g)
In an equilibrium mixture, the partial pressures are
P$$_{SO_3}$$ = 43 kPa; P$$_{O_2}$$ = 530 Pa and
P$$_{SO_2}$$ = 45 kPa. The equilibrium constant K$$_P$$ = ___ $$\times 10^{-2}$$. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 20th July 2021 Shift 1 - Chemistry - Question 55
250 mL of 0.5M NaOH was added to 500 mL of 1M HCl. The number of unreacted HCl molecules in the solution is p$$\times 10^{21}$$. Find out p.
(Nearest integer) (N$$_A$$ = 6.022 $$\times 10^{23}$$)
789
456
123
0.-
Clear All
NTA JEE Main 20th July 2021 Shift 1 - Chemistry - Question 56
At 20$$^\circ$$C, the vapour pressure of benzene is 70 torr and that of methyl benzene is 20 torr. The mole fraction of benzene in the vapour phase at 20$$^\circ$$C above an equimolar mixture of benzene and methyl benzene is ___ $$\times 10^{-2}$$. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 20th July 2021 Shift 1 - Chemistry - Question 57
The inactivation rate of a viral preparation is proportional to the amount of virus. In the first minute after preparation, 10% of the virus is inactivated. The rate constant for viral inactivation is ___ $$\times 10^{-3}$$ min$$^{-1}$$.
(Nearest integer) [Use : ln 10 = 2.303; $$\log_{10} 3 = 0.477$$ property of logarithm: $$\log x^y = y \log x$$]
789
456
123
0.-
Clear All
NTA JEE Main 20th July 2021 Shift 1 - Chemistry - Question 58
The spin-only magnetic moment value for the complex [Co(CN)$$_6$$]$$^{4-}$$ is ___ BM. (nearest integer value)
[At. no. of Co = 27]
789
456
123
0.-
Clear All
NTA JEE Main 20th July 2021 Shift 1 - Chemistry - Question 59
To synthesise 1 mole of 2-methylpropan-2-ol from Ethylethanoate equivalents of CH$$_3$$MgBr reagent will be required. (Integer value)
789
456
123
0.-
Clear All
NTA JEE Main 20th July 2021 Shift 1 - Chemistry - Question 60
The number of nitrogen atoms in a semicarbazone molecule of acetone is ___.
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




