NTA JEE Main 17th March 2021 Shift 1
For the following questions answer them individually
NTA JEE Main 17th March 2021 Shift 1 - Question 51
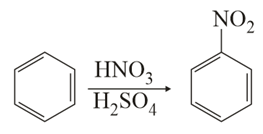
In the above reaction, 3.9 g of benzene on nitration gives 4.92 g of nitrobenzene. The percentage yield of nitrobenzene in the above reaction is ________ %. (Round off to the Nearest Integer).
(Given atomic mass: C: 12.0 u, H: 1.0 u, O: 16.0 u, N: 14.0 u)
789
456
123
0.-
Clear All
NTA JEE Main 17th March 2021 Shift 1 - Question 52
The mole fraction of a solute in a 100 molal aqueous solution is ________ $$\times 10^{-2}$$. (Round off to the Nearest Integer).
[Given: Atomic masses: H: 1.0 u, O: 16.0 u]
789
456
123
0.-
Clear All
NTA JEE Main 17th March 2021 Shift 1 - Question 53
A certain orbital has $$n = 4$$ and $$m_l = -3$$. The number of radial nodes in this orbital is ________. (Round off to the Nearest Integer).
789
456
123
0.-
Clear All
NTA JEE Main 17th March 2021 Shift 1 - Question 54
The pressure exerted by a non-reactive gaseous mixture of 6.4 g of methane and 8.8 g of carbon dioxide in a 10 L vessel at 27°C is ________ kPa. (Round off to the Nearest Integer).
[Assume gases are ideal, R = 8.314 J mol$$^{-1}$$ K$$^{-1}$$, Atomic masses: C: 12.0 u, H: 1.0 u, O: 16.0 u]
789
456
123
0.-
Clear All
NTA JEE Main 17th March 2021 Shift 1 - Question 55
The standard enthalpies of formation of $$Al_2O_3$$ and CaO are $$-1675$$ kJ mol$$^{-1}$$ and $$-635$$ kJ mol$$^{-1}$$ respectively. For the reaction $$3CaO + 2Al \to 3Ca + Al_2O_3$$ the standard reaction enthalpy $$\Delta_r H^0$$ = ________ kJ. (Round off to the Nearest Integer).
789
456
123
0.-
Clear All
NTA JEE Main 17th March 2021 Shift 1 - Question 56
0.01 moles of a weak acid HA ($$K_a = 2.0 \times 10^{-6}$$) is dissolved in 1.0 L of 0.1M HCl solution. The degree of dissociation of HA is ________ $$\times 10^{-5}$$. (Round off to the Nearest Integer). [Neglect volume change on adding HA and assume degree of dissociation << 1]
789
456
123
0.-
Clear All
NTA JEE Main 17th March 2021 Shift 1 - Question 57
15 mL of aqueous solution of $$Fe^{2+}$$ in acidic medium completely reacted with 20 mL of 0.03 M aqueous $$Cr_2O_7^{2-}$$. The molarity of the $$Fe^{2+}$$ solution is ________ $$\times 10^{-2}$$ M. (Round off to the Nearest Integer).
789
456
123
0.-
Clear All
NTA JEE Main 17th March 2021 Shift 1 - Question 58
The oxygen dissolved in water exerts a partial pressure of 20 kPa in the vapour above water. The molar solubility of oxygen in water is ________ $$\times 10^{-5}$$ mol dm$$^{-3}$$. (Round off to the Nearest Integer).
[Given: Henry's law constant = $$K_H = 8.0 \times 10^4$$ kPa for O$$_2$$. Density of water with dissolved oxygen = 1.0 kg dm$$^{-3}$$]
789
456
123
0.-
Clear All
NTA JEE Main 17th March 2021 Shift 1 - Question 59
For a certain first order reaction 32% of the reactant is left after 570 s. The rate constant of this reaction is ________ $$\times 10^{-3}$$ s$$^{-1}$$. (Round off to the Nearest Integer).
[Given: $$\log_{10} 2 = 0.301$$, $$\ln 10 = 2.303$$]
789
456
123
0.-
Clear All
NTA JEE Main 17th March 2021 Shift 1 - Question 60
The reaction of white phosphorus on boiling with alkali in inert atmosphere resulted in the formation of product A. The reaction 1 mol of A with excess of AgNO$$_3$$ in aqueous medium gives ________ mole(s) of Ag. (Round off to the Nearest Integer).
789
456
123
0.-
Clear All


