NTA JEE Main 16th March 2021 Shift 2
For the following questions answer them individually
NTA JEE Main 16th March 2021 Shift 2 - Question 31
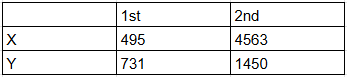
Identify the elements X and Y using the ionization energy values given below:
Ionization energy (kJ/mol):
NTA JEE Main 16th March 2021 Shift 2 - Question 32
The characteristics of elements X, Y and Z with atomic numbers, respectively, 33, 53 and 83 are:
NTA JEE Main 16th March 2021 Shift 2 - Question 33
Statement I: Sodium hydride can be used as an oxidising agent.
Statement II: The lone pair of electrons on nitrogen in pyridine makes it basic.
Choose the CORRECT answer from the options given below:
NTA JEE Main 16th March 2021 Shift 2 - Question 34
The exact volumes of 1 M NaOH solution required to neutralise 50 mL of 1 M $$H_3PO_3$$ solution and 100 mL of 2 M $$H_3PO_2$$ solution, respectively, are:
NTA JEE Main 16th March 2021 Shift 2 - Question 35
The correct statements about $$H_2O_2$$ are:
(A) used in the treatment of effluents.
(B) used as both oxidising and reducing agents.
(C) the two hydroxyl groups lie in the same plane.
(D) miscible with water.
Choose the correct answer from the options given below:
NTA JEE Main 16th March 2021 Shift 2 - Question 36
The INCORRECT statement regarding the structure of $$C_{60}$$ is:
NTA JEE Main 16th March 2021 Shift 2 - Question 37
An unsaturated hydrocarbon X on ozonolysis gives A. Compound A when warmed with ammoniacal silver nitrate forms a bright silver mirror along the sides of the test tube. The unsaturated hydrocarbon X is:
NTA JEE Main 16th March 2021 Shift 2 - Question 38
The green house gas/es is (are):
(A) Carbon dioxide
(B) Oxygen
(C) Water vapour
(D) Methane
Choose the most appropriate answer from the options given below:
NTA JEE Main 16th March 2021 Shift 2 - Question 39
The INCORRECT statements below regarding colloidal solutions is:
NTA JEE Main 16th March 2021 Shift 2 - Question 40
Which of the following reduction reaction CANNOT be carried out with coke?


