NTA JEE Main 16th March 2021 Shift 1
For the following questions answer them individually
NTA JEE Main 16th March 2021 Shift 1 - Question 31
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R:
Assertion A: The H-O-H bond angle in water molecule is 104.5°.
Reason R: The lone pair - lone pair repulsion of electrons is higher than the bond pair - bond pair repulsion.
NTA JEE Main 16th March 2021 Shift 1 - Question 32
Given below are two statements:
Statement I: $$H_2O_2$$ can act as both oxidising and reducing agent in basic medium.
Statement II: In the hydrogen economy, the energy is transmitted in the form of dihydrogen. In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Main 16th March 2021 Shift 1 - Question 33
A group 15 element, which is a metal and forms a hydride with strongest reducing power among group 15 hydrides. The element is:
NTA JEE Main 16th March 2021 Shift 1 - Question 34
Given below are two statements:
Statement I: Both $$CaCl_2 \cdot 6H_2O$$ and $$MgCl_2 \cdot 8H_2O$$ undergo dehydration on heating.
Statement II: BeO is amphoteric, whereas the oxides of other elements in the same group are acidic.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Main 16th March 2021 Shift 1 - Question 35
Among the following, the aromatic compounds are:
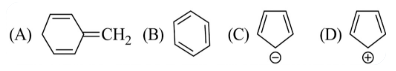
Choose the correct answer from the following options:
NTA JEE Main 16th March 2021 Shift 1 - Question 36
Assertion A: Enol form of acetone [$$CH_3COCH_3$$] exists in < 0.1% quantity. However, the enol form of acetyl acetone [$$CH_3COCH_2OCCH_3$$] exists in approximately 15% quantity.
Reason R: enol form of acetyl acetone is stabilized by intramolecular hydrogen bonding, which is not possible in enol form of acetone.
Choose the correct statement:
NTA JEE Main 16th March 2021 Shift 1 - Question 37
In chromatography technique, the purification of a compound is independent of:
NTA JEE Main 16th March 2021 Shift 1 - Question 38
The product "A" and "B" formed in below reactions are: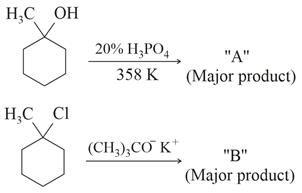
NTA JEE Main 16th March 2021 Shift 1 - Question 39
The type of pollution that gets increased during the daytime and in the presence of $$O_3$$ is:
NTA JEE Main 16th March 2021 Shift 1 - Question 40
Given below are two statements:
Statement I: The E° value of $$Ce^{4+}/Ce^{3+}$$ is +1.74 V.
Statement II: Ce is more stable in $$Ce^{4+}$$ state than $$Ce^{3+}$$ state.
In the light of the above statements, choose the most appropriate answer from the options given below:


