NTA JEE Main 15th April 2018 Shift 1
For the following questions answer them individually
NTA JEE Main 15th April 2018 Shift 1 - Question 41
In which of the following reactions, an increase in the volume of the container will favour the formation of products?
NTA JEE Main 15th April 2018 Shift 1 - Question 42
Which of the following is a Lewis acid?
NTA JEE Main 15th April 2018 Shift 1 - Question 43
The minimum volume of water required to dissolve 0.1 g lead (II) chloride to get a saturated solution ($$K_{SP}$$ of PbCl$$_2$$ = $$3.2 \times 10^{-8}$$; atomic mass of Pb = 207 u) is:
NTA JEE Main 15th April 2018 Shift 1 - Question 44
A white sodium salt dissolves readily in water to give a solution which is neutral to litmus. When silver nitrate solution is added to the aforementioned solution, a white precipitate is obtained which does not dissolve in dil. nitric acid. The anion is:
NTA JEE Main 15th April 2018 Shift 1 - Question 45
The correct match between items of List-I and List-II is:

NTA JEE Main 15th April 2018 Shift 1 - Question 46
The IUPAC name of the following compound is: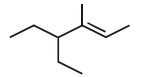
NTA JEE Main 15th April 2018 Shift 1 - Question 47
Which of the following will most readily give the dehydrohalogenation product?
NTA JEE Main 15th April 2018 Shift 1 - Question 48
The major product of the following reaction is: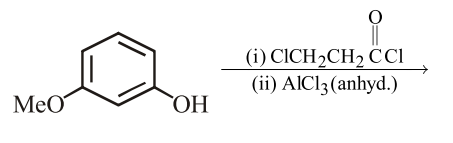
NTA JEE Main 15th April 2018 Shift 1 - Question 49
The increasing order of nitration of the following compounds is: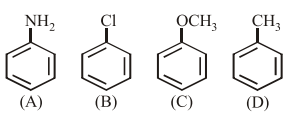
(A) Aniline, (B) Chlorobenzene, (C) Anisole, (D) Toluene
NTA JEE Main 15th April 2018 Shift 1 - Question 50
Which of the following arrangements shows the schematic alignment of magnetic moments of antiferromagnetic substance?


