NTA JEE Main 15th April 2018 Shift 1
For the following questions answer them individually
NTA JEE Main 15th April 2018 Shift 1 - Question 31
A sample of NaClO$$_3$$ is converted by heat to NaCl with a loss of 0.16 g of oxygen. The residue is dissolved in water and precipitated as AgCl. The mass of AgCl (in g) obtained will be: (Given: Molar mass of AgCl = 143.5 g mol$$^{-1}$$)
NTA JEE Main 15th April 2018 Shift 1 - Question 32
Ejection of the photoelectron from metal in the photoelectric effect experiment can be stopped by applying 0.5 V when the radiation of 250 nm is used. The work function of the metal is:
NTA JEE Main 15th April 2018 Shift 1 - Question 33
For Na$$^+$$, Mg$$^{2+}$$, F$$^-$$ and O$$^{2-}$$; the correct order of increasing ionic radii is:
NTA JEE Main 15th April 2018 Shift 1 - Question 34
In the molecular orbital diagram for the molecular ion, N$$_2^+$$, the number of electrons in the $$\sigma_{2p}$$ molecular orbital is:
NTA JEE Main 15th April 2018 Shift 1 - Question 35
Identify the pair in which the geometry of the species is T-shape and square pyramidal, respectively:
NTA JEE Main 15th April 2018 Shift 1 - Question 36
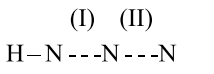
In hydrogen azide, the bond orders of bonds (I) and (II) are:
NTA JEE Main 15th April 2018 Shift 1 - Question 37
In graphite and diamond, the percentage of p characters of the hybrid orbitals in hybridization are respectively:
NTA JEE Main 15th April 2018 Shift 1 - Question 38
The decreasing order of bond angles in BF$$_3$$, NH$$_3$$, PF$$_3$$ and I$$_3^-$$ is:
NTA JEE Main 15th April 2018 Shift 1 - Question 39
For which of the following reactions, $$\Delta H$$ is equal to $$\Delta U$$?
NTA JEE Main 15th April 2018 Shift 1 - Question 40
An ideal gas undergoes a cyclic process as shown in Figure.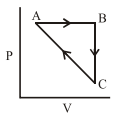
$$\Delta U_{BC} = -5$$ kJ mol$$^{-1}$$, $$q_{AB} = 2$$ kJ mol$$^{-1}$$
$$W_{AB} = -5$$ kJ mol$$^{-1}$$, $$W_{CA} = 3$$ kJ mol$$^{-1}$$
Heat absorbed by the system during process CA is:


