NTA JEE Main 13th April 2023 Shift 2
For the following questions answer them individually
NTA JEE Main 13th April 2023 Shift 2 - Question 41
Given below are two statements, one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A : The diameter of colloidal particles in solution should not be much smaller than wavelength of light to show Tyndall effect.
Reason R : The light scatters in all directions when the size of particles is large enough.
In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Main 13th April 2023 Shift 2 - Question 42
Given below are two statements related to Ellingham diagram :
Statement I : Ellingham diagrams can be constructed for formation of oxides, sulphides and halides of metals.
Statement II : It consists of plots of $$\Delta H°$$ vs T for formation of oxides of elements.
In the light of the above statements, choose the most appropriate answer from the options given below :
NTA JEE Main 13th April 2023 Shift 2 - Question 43
The correct group of halide ions which can be oxidised by oxygen in acidic medium is
NTA JEE Main 13th April 2023 Shift 2 - Question 44
The covalency and oxidation state respectively of boron in BF$$_4^-$$, are
NTA JEE Main 13th April 2023 Shift 2 - Question 45
Which of the following complexes will exhibit maximum attraction to an applied magnetic field?
NTA JEE Main 13th April 2023 Shift 2 - Question 46
The total number of stereoisomers for the complex $$[Cr(ox)_2ClBr]^{3-}$$ (where ox = oxalate) is
NTA JEE Main 13th April 2023 Shift 2 - Question 47
Match List-I with List-II.
1-Bromopropane is reacted with reagents in List-I to give product in List-II
| List-I Reagent | List-II Product | ||
|---|---|---|---|
| A | KOH (alc) | I | Nitrile |
| B | KCN (alc) | II | Ester |
| C | AgNO$$_2$$ | III | Alkene |
| D | H$$_3$$CCOOAg | IV | Nitroalkane |
Choose the correct answer from the options given below
NTA JEE Main 13th April 2023 Shift 2 - Question 48
Compound A from the following reaction sequence is :
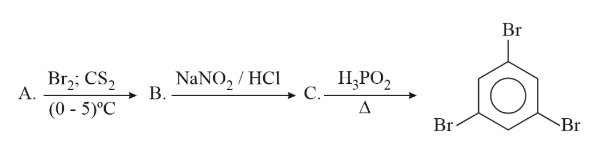
NTA JEE Main 13th April 2023 Shift 2 - Question 49
In the wet tests for detection of various cations by precipitation, Ba$$^{2+}$$ cations are detected by obtaining precipitate of
NTA JEE Main 13th April 2023 Shift 2 - Question 50
The naturally occurring amino acid that contains only one basic functional group in its chemical structure is


