NTA JEE Main 13th April 2023 Shift 2
For the following questions answer them individually
NTA JEE Main 13th April 2023 Shift 2 - Question 31
Identify the correct order of standard enthalpy of formation of sodium halides.
NTA JEE Main 13th April 2023 Shift 2 - Question 32
Match List-I with List-II.
| List-I | List-II | ||
|---|---|---|---|
| A | Weak intermolecular forces of attraction | I | Hexamethylenediamine + adipic acid |
| B | Hydrogen bonding | II | AlEt$$_3$$ + TiCl$$_4$$ |
| C | Heavily branched polymer | III | 2-chloro-1, 3-butadiene |
| D | High density polymer | IV | Phenol + formaldehyde |
Choose the correct answer from the options given below
NTA JEE Main 13th April 2023 Shift 2 - Question 33
Given below are two statements :
Statement I : SO$$_2$$ and H$$_2$$O both possess V-shaped structure
Statement II : The bond angle of SO$$_2$$ is less than that of H$$_2$$O.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 13th April 2023 Shift 2 - Question 34
What happens when methane undergoes combustion in systems A and B respectively?
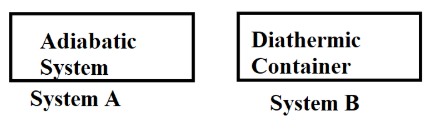
NTA JEE Main 13th April 2023 Shift 2 - Question 35
Given below are two statements, one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A : Isotopes of hydrogen have almost same chemical properties, but difference in their rates of reactions.
Reason R: Isotopes of hydrogen have different enthalpy of bond dissociation.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 13th April 2023 Shift 2 - Question 36
Better method for preparation of BeF$$_2$$, among the following is
NTA JEE Main 13th April 2023 Shift 2 - Question 37
The major product for the following reaction is :
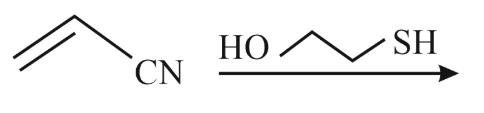
NTA JEE Main 13th April 2023 Shift 2 - Question 38
Given below are two statements, one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A : Order of acidic nature of the following compounds is A > B > C.
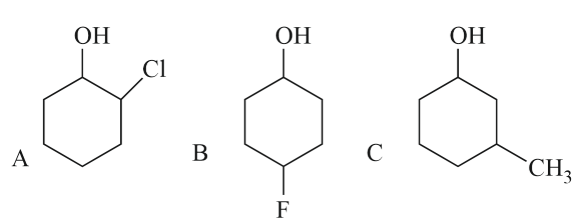
Reason R: Fluoro is a stronger electron withdrawing group than Chloro group.
In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Main 13th April 2023 Shift 2 - Question 39
Given below are two statements :
Statement I : Tropolone is an aromatic compound and has $$8\pi$$ electrons.
Statement II : $$\pi$$ electrons of $$> C = O$$ group in tropolone is involved in aromaticity. In the light of the above statements choose the correct answer from the options given below:
NTA JEE Main 13th April 2023 Shift 2 - Question 40
Which of the following are the Green house gases?
A. Water vapour
B. Ozone
C. I$$_2$$
D. Molecular hydrogen
Choose the most appropriate answer from the options given below :


