NTA JEE Main 12th January 2019 Shift 2
For the following questions answer them individually
NTA JEE Main 12th January 2019 Shift 2 - Question 41
The major product of the following reaction is:
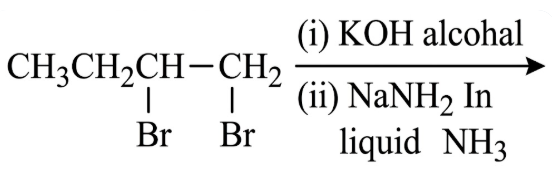
NTA JEE Main 12th January 2019 Shift 2 - Question 42
The major product of the following reaction is: (compound with Cl reacting with NaOEt/heat)
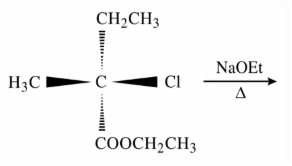
NTA JEE Main 12th January 2019 Shift 2 - Question 43
The compound that is NOT a common component of photochemical smog is:
NTA JEE Main 12th January 2019 Shift 2 - Question 44
The upper stratosphere consisting of the ozone layer, protects us from the sun's radiation that falls in the wavelength region of
NTA JEE Main 12th January 2019 Shift 2 - Question 45
Molecules of benzoic acid ($$C_6H_5COOH$$) dimerise in 30 g of benzene. 'w' g of benzoic acid shows a depression in freezing point equal to 2 K. If the percentage association of the acid to form dimer in the solution is 80, then w is: (Given that $$K_f = 5$$ K mol$$^{-1}$$, molar mass of benzoic acid = 122 g mol$$^{-1}$$)
NTA JEE Main 12th January 2019 Shift 2 - Question 46
$$\Lambda_m^°$$ for NaCl, HCl and NaA are 126.4, 425.9 and 100.5 S cm$$^2$$ mol$$^{-1}$$ respectively. If the conductivity of 0.001 M HA is $$5 \times 10^{-5}$$ S cm$$^{-1}$$, degree of dissociation of HA is
NTA JEE Main 12th January 2019 Shift 2 - Question 47
For a reaction, consider the plot of ln k versus 1/T given in the figure. If the rate constant of this reaction at 400 K is $$10^{-5} s^{-1}$$, then the rate constant at 500 K is:
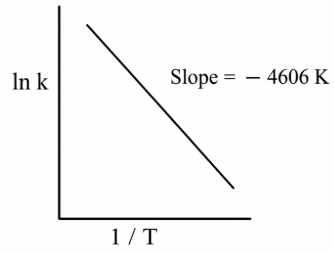
NTA JEE Main 12th January 2019 Shift 2 - Question 48
Among the following, the false statement is:
NTA JEE Main 12th January 2019 Shift 2 - Question 49
The pair that does not require calcination is
NTA JEE Main 12th January 2019 Shift 2 - Question 50
Chlorine on reaction with hot and concentrated sodium hydroxide gives


