NTA JEE Main 12th January 2019 Shift 2
For the following questions answer them individually
NTA JEE Main 12th January 2019 Shift 2 - Question 31
8 g of NaOH is dissolved in 18g of $$H_2O$$. Mole fraction of NaOH in solution and molality (in mol kg$$^{-1}$$) of the solution respectively are:
NTA JEE Main 12th January 2019 Shift 2 - Question 32
If the de Broglie wavelength of the electron in $$n^{th}$$ Bohr orbit in a hydrogenic atom is equal to $$1.5\pi a_0$$ ($$a_0$$ is Bohr radius), then the value of $$\frac{n}{z}$$ is:
NTA JEE Main 12th January 2019 Shift 2 - Question 33
The element that does not show catenation is
NTA JEE Main 12th January 2019 Shift 2 - Question 34
The correct order of atomic radii is:
NTA JEE Main 12th January 2019 Shift 2 - Question 35
The element that shows greater ability to form $$p\pi - p\pi$$ multiple bonds is:
NTA JEE Main 12th January 2019 Shift 2 - Question 36
An open vessel at $$27°C$$ is heated until two fifth of the air (assumed as an ideal gas) in it has escaped from the vessel. Assuming that the volume of the vessel remains constant, the temperature to which the vessel has heated is:
NTA JEE Main 12th January 2019 Shift 2 - Question 37
Given:
(i) C(graphite) + O$$_2$$(g) $$\to$$ CO$$_2$$(g); $$\Delta_r H^{\ominus} = x$$ kJ mol$$^{-1}$$,
(ii) C(graphite) + $$\frac{1}{2}$$O$$_2$$(g) $$\to$$ CO(g); $$\Delta_r H^{\ominus} = y$$ kJ mol$$^{-1}$$,
(iii) CO(g) + $$\frac{1}{2}$$O$$_2$$(g) $$\to$$ CO$$_2$$(g); $$\Delta_r H^{\ominus} = z$$ kJ mol$$^{-1}$$.
Based on the above thermochemical equations, find out which one of the following algebraic relationships is correct?
NTA JEE Main 12th January 2019 Shift 2 - Question 38
The combination of plots which does not represent isothermal expansion of an ideal gas is
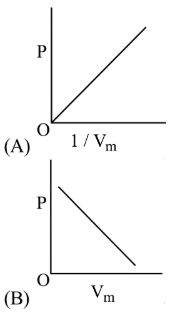
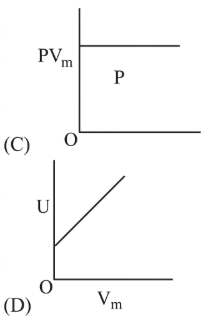
NTA JEE Main 12th January 2019 Shift 2 - Question 39
If $$K_{sp}$$ of $$Ag_2CO_3$$ is $$8 \times 10^{-12}$$, the molar solubility of $$Ag_2CO_3$$ in 0.1 M $$AgNO_3$$ is:
NTA JEE Main 12th January 2019 Shift 2 - Question 40
The volume strength of 1M $$H_2O_2$$ is: (Molar mass of $$H_2O_2 = 34$$ g mol$$^{-1}$$)


