NTA JEE Main 12th January 2019 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 12th January 2019 Shift 2 - Chemistry - Question 31
8 g of NaOH is dissolved in 18g of $$H_2O$$. Mole fraction of NaOH in solution and molality (in mol kg$$^{-1}$$) of the solution respectively are:
NTA JEE Main 12th January 2019 Shift 2 - Chemistry - Question 32
If the de Broglie wavelength of the electron in $$n^{th}$$ Bohr orbit in a hydrogenic atom is equal to $$1.5\pi a_0$$ ($$a_0$$ is Bohr radius), then the value of $$\frac{n}{z}$$ is:
NTA JEE Main 12th January 2019 Shift 2 - Chemistry - Question 33
The element that does not show catenation is
NTA JEE Main 12th January 2019 Shift 2 - Chemistry - Question 34
The correct order of atomic radii is:
NTA JEE Main 12th January 2019 Shift 2 - Chemistry - Question 35
The element that shows greater ability to form $$p\pi - p\pi$$ multiple bonds is:
NTA JEE Main 12th January 2019 Shift 2 - Chemistry - Question 36
An open vessel at $$27°C$$ is heated until two fifth of the air (assumed as an ideal gas) in it has escaped from the vessel. Assuming that the volume of the vessel remains constant, the temperature to which the vessel has heated is:
NTA JEE Main 12th January 2019 Shift 2 - Chemistry - Question 37
Given:
(i) C(graphite) + O$$_2$$(g) $$\to$$ CO$$_2$$(g); $$\Delta_r H^{\ominus} = x$$ kJ mol$$^{-1}$$,
(ii) C(graphite) + $$\frac{1}{2}$$O$$_2$$(g) $$\to$$ CO(g); $$\Delta_r H^{\ominus} = y$$ kJ mol$$^{-1}$$,
(iii) CO(g) + $$\frac{1}{2}$$O$$_2$$(g) $$\to$$ CO$$_2$$(g); $$\Delta_r H^{\ominus} = z$$ kJ mol$$^{-1}$$.
Based on the above thermochemical equations, find out which one of the following algebraic relationships is correct?
NTA JEE Main 12th January 2019 Shift 2 - Chemistry - Question 38
The combination of plots which does not represent isothermal expansion of an ideal gas is
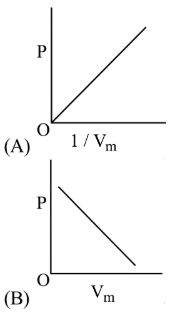
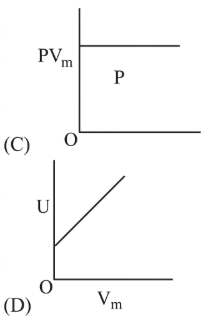
NTA JEE Main 12th January 2019 Shift 2 - Chemistry - Question 39
If $$K_{sp}$$ of $$Ag_2CO_3$$ is $$8 \times 10^{-12}$$, the molar solubility of $$Ag_2CO_3$$ in 0.1 M $$AgNO_3$$ is:
NTA JEE Main 12th January 2019 Shift 2 - Chemistry - Question 40
The volume strength of 1M $$H_2O_2$$ is: (Molar mass of $$H_2O_2 = 34$$ g mol$$^{-1}$$)
NTA JEE Main 12th January 2019 Shift 2 - Chemistry - Question 41
The major product of the following reaction is:
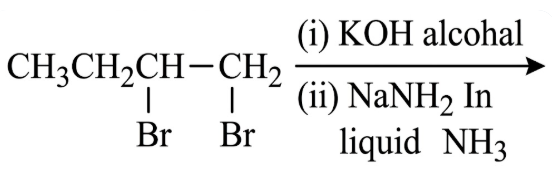
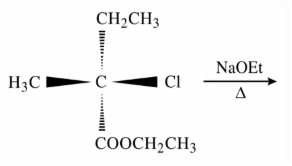
NTA JEE Main 12th January 2019 Shift 2 - Chemistry - Question 42
The major product of the following reaction is: (compound with Cl reacting with NaOEt/heat)
NTA JEE Main 12th January 2019 Shift 2 - Chemistry - Question 43
The compound that is NOT a common component of photochemical smog is:
NTA JEE Main 12th January 2019 Shift 2 - Chemistry - Question 44
The upper stratosphere consisting of the ozone layer, protects us from the sun's radiation that falls in the wavelength region of
NTA JEE Main 12th January 2019 Shift 2 - Chemistry - Question 45
Molecules of benzoic acid ($$C_6H_5COOH$$) dimerise in 30 g of benzene. 'w' g of benzoic acid shows a depression in freezing point equal to 2 K. If the percentage association of the acid to form dimer in the solution is 80, then w is: (Given that $$K_f = 5$$ K mol$$^{-1}$$, molar mass of benzoic acid = 122 g mol$$^{-1}$$)
NTA JEE Main 12th January 2019 Shift 2 - Chemistry - Question 46
$$\Lambda_m^°$$ for NaCl, HCl and NaA are 126.4, 425.9 and 100.5 S cm$$^2$$ mol$$^{-1}$$ respectively. If the conductivity of 0.001 M HA is $$5 \times 10^{-5}$$ S cm$$^{-1}$$, degree of dissociation of HA is
NTA JEE Main 12th January 2019 Shift 2 - Chemistry - Question 47
For a reaction, consider the plot of ln k versus 1/T given in the figure. If the rate constant of this reaction at 400 K is $$10^{-5} s^{-1}$$, then the rate constant at 500 K is:
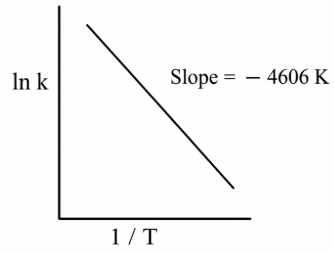
NTA JEE Main 12th January 2019 Shift 2 - Chemistry - Question 48
Among the following, the false statement is:
NTA JEE Main 12th January 2019 Shift 2 - Chemistry - Question 49
The pair that does not require calcination is
NTA JEE Main 12th January 2019 Shift 2 - Chemistry - Question 50
Chlorine on reaction with hot and concentrated sodium hydroxide gives
NTA JEE Main 12th January 2019 Shift 2 - Chemistry - Question 51
The magnetic moment of an octahedral homoleptic Mn(II) complex is 5.9 B.M. The suitable ligand for this complex is:
NTA JEE Main 12th January 2019 Shift 2 - Chemistry - Question 52
The major product of the following reaction is:
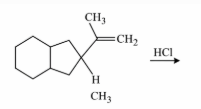
NTA JEE Main 12th January 2019 Shift 2 - Chemistry - Question 53
The major product in the following conversion is:
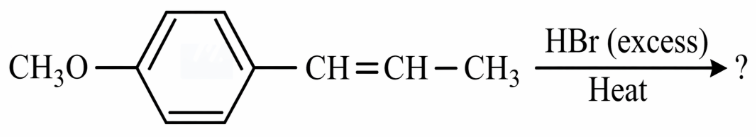
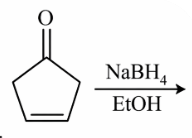
NTA JEE Main 12th January 2019 Shift 2 - Chemistry - Question 54
The major product of the following reaction is:
NTA JEE Main 12th January 2019 Shift 2 - Chemistry - Question 55
The aldehydes which will not form Grignard product with one equivalent of Grignard reagents are:
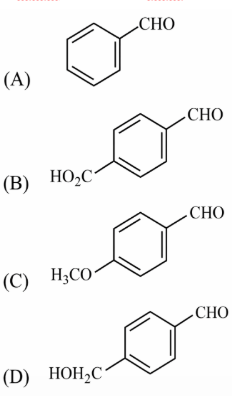
NTA JEE Main 12th January 2019 Shift 2 - Chemistry - Question 56
The increasing order of the reactivity of the following with LiAlH$$_4$$ is:
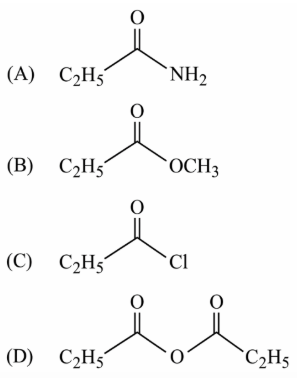
NTA JEE Main 12th January 2019 Shift 2 - Chemistry - Question 57
The major product of the following reaction is:
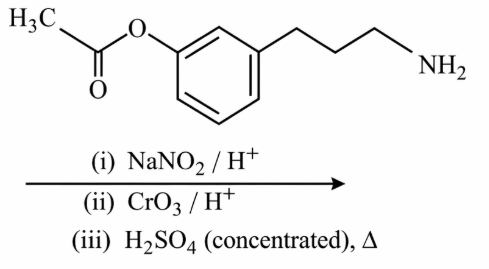
NTA JEE Main 12th January 2019 Shift 2 - Chemistry - Question 58
The two monomers for the synthesis of nylon 6,6 are
NTA JEE Main 12th January 2019 Shift 2 - Chemistry - Question 59
The correct statement(s) among I to III with respect to potassium ions that are abundant within the cell fluids, is/are: I. They activate many enzymes. II. They participate in the oxidation of glucose to produce ATP. III. Along with sodium ions, they are responsible for the transmission of nerve signals.
NTA JEE Main 12th January 2019 Shift 2 - Chemistry - Question 60
The correct structure of histidine in a strongly acidic solution (pH = 2) is
.webp)
.webp)
.webp)
.webp)




