NTA JEE Main 12th April 2019 Shift 2
For the following questions answer them individually
NTA JEE Main 12th April 2019 Shift 2 - Question 31
25 g of an unknown hydrocarbon upon burning produces 88 g of CO$$_2$$ and 9 g of H$$_2$$O. This unknown hydrocarbon contains:
NTA JEE Main 12th April 2019 Shift 2 - Question 32
Among the following, the energy of 2s orbital is lowest in:
NTA JEE Main 12th April 2019 Shift 2 - Question 33
In comparison to boron, beryllium has:
NTA JEE Main 12th April 2019 Shift 2 - Question 34
The incorrect match in the following is:
NTA JEE Main 12th April 2019 Shift 2 - Question 35
In which one of the following equilibria, K$$_p$$ ≠ K$$_c$$?
NTA JEE Main 12th April 2019 Shift 2 - Question 36
The molar solubility of Cd(OH)$$_2$$ is $$1.84 \times 10^{-5}$$ M in water. The expected solubility of Cd(OH)$$_2$$ in a buffer solution of pH = 12 is:
NTA JEE Main 12th April 2019 Shift 2 - Question 37
The temporary hardness of a water sample is due to compound X. Boiling this sample converts X to compound Y. X and Y, respectively, are:
NTA JEE Main 12th April 2019 Shift 2 - Question 38
The incorrect statement is:
NTA JEE Main 12th April 2019 Shift 2 - Question 39
The C - C bond length is maximum in:
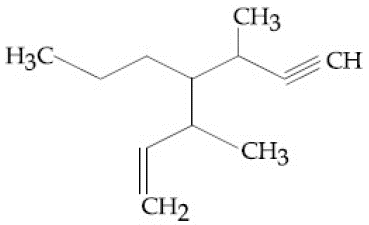
NTA JEE Main 12th April 2019 Shift 2 - Question 40
The IUPAC name for the following compound is: