NTA JEE Main 12th April 2019 Shift 1
For the following questions answer them individually
NTA JEE Main 12th April 2019 Shift 1 - Question 31
5 moles of AB$$_2$$ weigh $$125 \times 10^{-3}$$ kg and 10 moles of A$$_2$$B$$_2$$ weigh $$300 \times 10^{-3}$$ kg. The molar mass of A (M$$_A$$) in kg mol$$^{-1}$$ are:
NTA JEE Main 12th April 2019 Shift 1 - Question 32
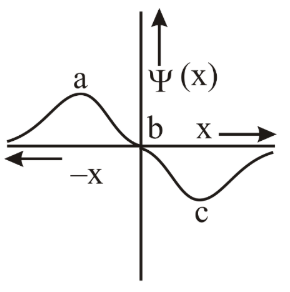
The electrons are more likely to be found:
NTA JEE Main 12th April 2019 Shift 1 - Question 33
The group number, number of valence electrons, and valency of an element with atomic number 15, respectively, are:
NTA JEE Main 12th April 2019 Shift 1 - Question 34
The correct statement among the following is:
NTA JEE Main 12th April 2019 Shift 1 - Question 35
An ideal gas is allowed to expand from 1 L to 10 L against a constant external pressure of 1 bar. The work done in kJ is:
NTA JEE Main 12th April 2019 Shift 1 - Question 36
Enthalpy of sublimation of iodine is 24 cal g$$^{-1}$$ at 200°C. If specific heat of I$$_2$$(s) and I$$_2$$(vap) are 0.055 and 0.031 cal g$$^{-1}$$ K$$^{-1}$$ respectively, then enthalpy of sublimation of iodine at 250°C in cal g$$^{-1}$$ is:
NTA JEE Main 12th April 2019 Shift 1 - Question 37
What is the molar solubility of Al(OH)$$_3$$ in 0.2 M NaOH solution? Given that, solubility product of Al(OH)$$_3$$ = $$2.4 \times 10^{-24}$$:
NTA JEE Main 12th April 2019 Shift 1 - Question 38
An example of a disproportionation reaction is
NTA JEE Main 12th April 2019 Shift 1 - Question 39
Given:
Co$$^{3+}$$ + e$$^-$$ $$\to$$ Co$$^{2+}$$; E° = +1.81V
Pb$$^{3+}$$ + 2e$$^-$$ $$\to$$ Pb$$^{2+}$$; E° = +1.67V
Ce$$^{4+}$$ + e$$^-$$ $$\to$$ Ce$$^{3+}$$; E° = +1.61V
Bi$$^{3+}$$ + 3e$$^-$$ $$\to$$ Bi; E° = +0.20V
Oxidizing power of the species will increase in the order:
NTA JEE Main 12th April 2019 Shift 1 - Question 40
The correct sequence of thermal stability of the following carbonates is:


