NTA JEE Main 10th April 2023 Shift 2
For the following questions answer them individually
NTA JEE Main 10th April 2023 Shift 2 - Question 31
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: 3.1500 g of hydrated oxalic acid dissolved in water to make 250.0 mL solution will result in 0.1 M oxalic acid solution.
Reason R: Molar mass of hydrated oxalic acid is 126 g mol$$^{-1}$$.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Main 10th April 2023 Shift 2 - Question 32
Match List-I with List-II
| List-I | List-II | ||
|---|---|---|---|
| A | 16 g of CH$$_4$$(g) | I | Weighs 28g |
| B | 1 g of H$$_2$$(g) | II | $$60.2 \times 10^{23}$$ electrons |
| C | 1 mole of N$$_2$$(g) | III | Weighs 32g |
| D | 0.5 mol of SO$$_2$$(g) | IV | Occupies 11.4 L volume at STP |
Choose the correct answer from the options given below:
NTA JEE Main 10th April 2023 Shift 2 - Question 33
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: The energy required to form Mg$$^{2+}$$ from Mg is much higher than that required to produce Mg$$^+$$
Reason R: Mg$$^{2+}$$ is small ion and carry more charge than Mg$$^+$$
In the light of the above statements, choose the correct answer from the options given below.
NTA JEE Main 10th April 2023 Shift 2 - Question 34
The correct order of metallic character is
NTA JEE Main 10th April 2023 Shift 2 - Question 35
Gibbs energy vs T plot for the formation of oxides is given below. For the given diagram, the correct statement is-
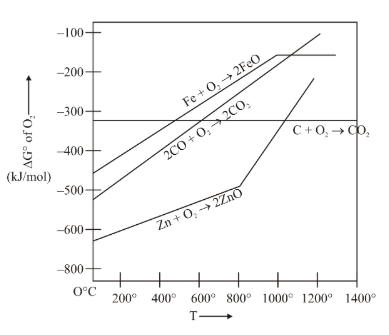
NTA JEE Main 10th April 2023 Shift 2 - Question 36
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: Physical properties of isotopes of hydrogen are different.
Reason R: Mass difference between isotopes of hydrogen is very large.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Main 10th April 2023 Shift 2 - Question 37
Number of water molecules in washing soda and soda ash respectively are:
NTA JEE Main 10th April 2023 Shift 2 - Question 38
The decreasing order of hydride affinity for following carbocations is:
(a)
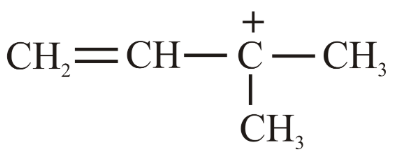
(b)
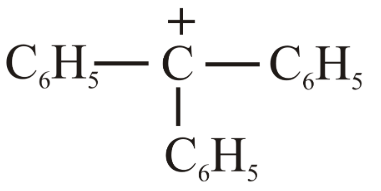
(c)
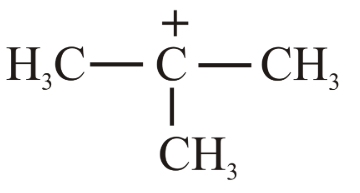
(d)
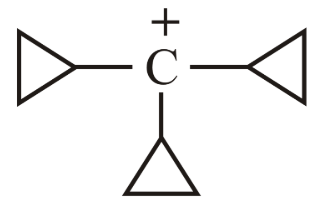
Choose the correct answer from the options given below:
NTA JEE Main 10th April 2023 Shift 2 - Question 39
The correct order for acidity of the following hydroxyl compound is
(A) CH$$_3$$OH
(B) (CH$$_3$$)$$_3$$COH
(C)
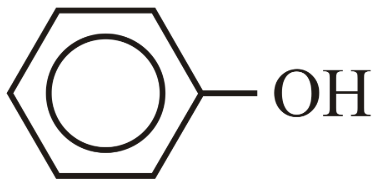
(D)
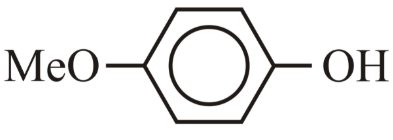
(E)
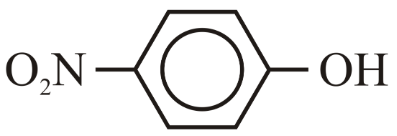
Choose the correct answer from the options given below:
NTA JEE Main 10th April 2023 Shift 2 - Question 40
In Carius tube, an organic compound 'X' is treated with sodium peroxide to form a mineral acid 'Y'. The solution of BaCl$$_2$$ is added to 'Y' to form a precipitate 'Z'. 'Z' is used for the quantitative estimation of an extra element. 'X' could be


