NTA JEE Main 10th April 2023 Shift 1
For the following questions answer them individually
NTA JEE Main 10th April 2023 Shift 1 - Question 31
The number of molecules and moles in 2.8375 litres of O$$_2$$ at STP are respectively
NTA JEE Main 10th April 2023 Shift 1 - Question 32
The pair from the following pairs having both compounds with net non-zero dipole moment is
NTA JEE Main 10th April 2023 Shift 1 - Question 33
The compound which does not exist is
NTA JEE Main 10th April 2023 Shift 1 - Question 34
The enthalpy change for the adsorption process and micelle formation respectively are
NTA JEE Main 10th April 2023 Shift 1 - Question 35
Given
(A) $$2CO(g) + O_2(g) \to 2CO_2(g)$$, $$\Delta H_1^0 = -x$$ kJ mol$$^{-1}$$
(B) $$C_{graphite} + O_2(g) \to CO_2(g)$$, $$\Delta H_2^0 = -y$$ kJ mol$$^{-1}$$
The $$\Delta H^0$$ for the reaction $$C_{graphite} + \frac{1}{2}O_2(g) \to CO(g)$$ is
NTA JEE Main 10th April 2023 Shift 1 - Question 36
Given below are two reactions, involved in the commercial production of dihydrogen H$$_2$$. The two reactions are carried out at temperature "T$$_1$$" and "T$$_2$$", respectively
$$C(s) + H_2O(g) \xrightarrow{T_1} CO(g) + H_2(g)$$
$$CO(g) + H_2O(g) \xrightarrow{T_2, Catalyst} CO_2(g) + H_2(g)$$
The temperatures T$$_1$$ and T$$_2$$ are correctly related as
NTA JEE Main 10th April 2023 Shift 1 - Question 37
Lime reacts exothermally with water to give 'A' which has low solubility in water. Aqueous solution of 'A' is often used for the test of CO$$_2$$, a test in which insoluble B is formed. If B is further reacted with CO$$_2$$ then soluble compound is formed. 'A' is
NTA JEE Main 10th April 2023 Shift 1 - Question 38
Using column chromatography, mixture of two compounds 'A' and 'B' was separated. 'A' eluted first, this indicates 'B' has
NTA JEE Main 10th April 2023 Shift 1 - Question 39
The major product 'P' formed in the given reaction is
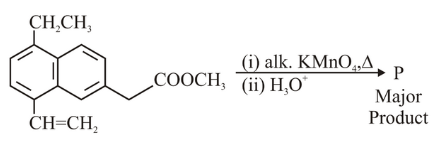
NTA JEE Main 10th April 2023 Shift 1 - Question 40
Match List I with List II
| List I Industry | List II Waste Generated | ||
|---|---|---|---|
| (A) | Steel plants | (I) | Gypsum |
| (B) | Thermal power plants | (II) | Fly ash |
| (C) | Fertilizer Industries | (III) | Slag |
| (D) | Paper mills | (IV) | Bio-degradable wastes |
Choose the correct answer from the options below:


