NTA JEE Main 10th April 2019 Shift 2
For the following questions answer them individually
NTA JEE Main 10th April 2019 Shift 2 - Question 41
The increasing order of nucleophilicity of the following nucleophiles is:
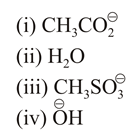
NTA JEE Main 10th April 2019 Shift 2 - Question 42
In chromatography, which of the following statements is incorrect for R$$_f$$?
NTA JEE Main 10th April 2019 Shift 2 - Question 43
Which of these factors does not govern the stability of a conformation in acyclic compounds?
NTA JEE Main 10th April 2019 Shift 2 - Question 44
The major product obtained in the given reaction is:
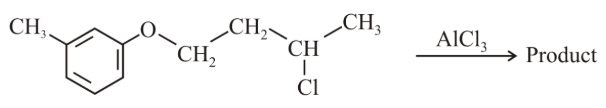
NTA JEE Main 10th April 2019 Shift 2 - Question 45
Air pollution that occurs in sunlight is
NTA JEE Main 10th April 2019 Shift 2 - Question 46
The noble gas that does not occur in the atmosphere is;
NTA JEE Main 10th April 2019 Shift 2 - Question 47
1 g of a non-volatile non-electrolyte solute is dissolved in 100 g of two different solvents A and B whose ebullioscopic constants are in the ratio of 1:5. The ratio of the elevation in their boiling points, $$\frac{\Delta T_{bA}}{\Delta T_{bB}}$$, is:
(assuming they have the same molar mass)
NTA JEE Main 10th April 2019 Shift 2 - Question 48
Which one of the following graphs between molar conductivity $$\Lambda_m$$ versus $$\sqrt{C}$$ is correct?
NTA JEE Main 10th April 2019 Shift 2 - Question 49
For the reaction of H$$_2$$ with I$$_2$$, the rate constant is $$2.5 \times 10^{-4}$$ dm$$^3$$ mol$$^{-1}$$ s$$^{-1}$$ at 327°C and 1.0 dm$$^3$$ mol$$^{-1}$$ s$$^{-1}$$ at 527°C. The activation energy for the reaction, in kJ mol$$^{-1}$$ is:
R = 8.314 JK$$^{-1}$$ mol$$^{-1}$$
NTA JEE Main 10th April 2019 Shift 2 - Question 50
The correct option among the following is.


