NTA JEE Main 10th April 2019 Shift 2
For the following questions answer them individually
NTA JEE Main 10th April 2019 Shift 2 - Question 31
The minimum amount of O$$_2$$(g) consumed per gram of reactant is for the reaction:
(Given atomic mass: Fe = 56, O = 16, Mg = 24, P = 31, C = 12, H = 1)
NTA JEE Main 10th April 2019 Shift 2 - Question 32
The ratio of the shortest wavelength of two spectral series of hydrogen spectrum is found to be about 9. The spectral series are:
NTA JEE Main 10th April 2019 Shift 2 - Question 33
The correct order of the first ionization enthalpies is:
NTA JEE Main 10th April 2019 Shift 2 - Question 34
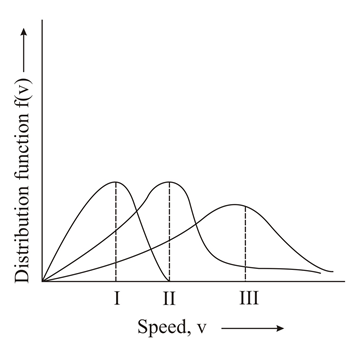
Points I, II and III in the following plot respectively correspond to (V$$_{mp}$$: most probable velocity)
NTA JEE Main 10th April 2019 Shift 2 - Question 35
The difference between ΔH and ΔU is ΔH - ΔU, when the combustion of one mole of heptane l is carried out at a temperature T, is equal to:
NTA JEE Main 10th April 2019 Shift 2 - Question 36
For the reaction,
2SO$$_2$$g + O$$_2$$g $$\rightleftharpoons$$ 2SO $$_3$$g,
ΔH = -57.2 kJ mol$$^{-1}$$ and K$$_c$$ = $$1.7 \times 10^{16}$$.
Which of the following statements is incorrect?
NTA JEE Main 10th April 2019 Shift 2 - Question 37
The pH of a 0.02 M NH$$_4$$Cl solution will be [Given: K$$_b$$ NH$$_4$$OH = 10$$^{-5}$$ and log 2 = 0.301]
NTA JEE Main 10th April 2019 Shift 2 - Question 38
The correct statements among a to d are:
a. Saline hydrides produce H$$_2$$ gas when reacted with H$$_2$$O.
b. Reaction of LiAlH$$_4$$ with BF$$_3$$ leads to B$$_2$$H$$_6$$.
c. PH$$_3$$ and CH$$_4$$ are electron-rich and electron-precise hydrides, respectively.
d. HF and CH$$_4$$ are called as molecular hydrides.
NTA JEE Main 10th April 2019 Shift 2 - Question 39
A hydrated solid X on heating initially gives a monohydrated compound Y. Y upon heating above 373 K leads to an anhydrous white powder Z. X and Z, respectively, are;
NTA JEE Main 10th April 2019 Shift 2 - Question 40
The number of pentagons in C$$_{60}$$ and trigons (triangles) in white phosphorus, are;


