NTA JEE Main 1st February 2023 Shift 2
For the following questions answer them individually
NTA JEE Main 1st February 2023 Shift 2 - Question 31
Which one of the following sets of ions represents a collection of isoelectronic species?
(Given: Atomic Number: F: 9, Cl: 17, Na = 11, Mg = 12, Al = 13, K = 19, Ca = 20, Sc = 21)
NTA JEE Main 1st February 2023 Shift 2 - Question 32
For electron gain enthalpies of the elements denoted as $$\Delta_{eg}$$H, the incorrect option is:
NTA JEE Main 1st February 2023 Shift 2 - Question 33
The effect of addition of helium gas to the following reaction in equilibrium state at constant volume, is:
$$\text{PCl}_5(g) \rightleftharpoons \text{PCl}_3(g) + \text{Cl}_2(g)$$
NTA JEE Main 1st February 2023 Shift 2 - Question 34
Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A): An aqueous solution of KOH when for volumetric analysis, its concentration should be checked before the use.
Reason (R): On aging, KOH solution absorbs atmospheric CO$$_2$$.
In the light of the above statements, choose the correct answer from the options given below.
NTA JEE Main 1st February 2023 Shift 2 - Question 35
O $$-$$ O bond length in H$$_2$$O$$_2$$ is X than the O $$-$$ O bond length in F$$_2$$O$$_2$$. The O $$-$$ H bond length in H$$_2$$O$$_2$$ is Y than that of the O $$-$$ F bond in F$$_2$$O$$_2$$. Choose the correct option for X and Y from the given below.
NTA JEE Main 1st February 2023 Shift 2 - Question 36
The starting material for convenient preparation of deuterated hydrogen peroxide (D$$_2$$O$$_2$$) in laboratory is:
NTA JEE Main 1st February 2023 Shift 2 - Question 37
Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A): Gypsum is used for making fireproof wall boards.
Reason (R): Gypsum is unstable at high temperatures.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Main 1st February 2023 Shift 2 - Question 38
The correct order of bond enthalpy (kJ mol$$^{-1}$$) is:
NTA JEE Main 1st February 2023 Shift 2 - Question 39
Given below are two statements:
Statement I: Sulphanilic acid gives esterification test for carboxyl group.
Statement II: Sulphanilic acid gives red colour in Lassigne's test for extra element detection.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 1st February 2023 Shift 2 - Question 40
Testosterone, which is a steroidal hormone, has the following structure.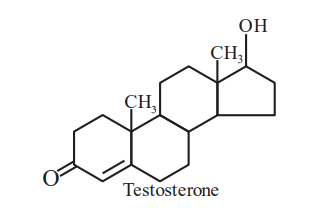
The total number of asymmetric carbon atom/s in testosterone is ______.


